Altamira Therapeutics Reports Positive Top-Line Data from Bentrio Clinical Trial in Seasonal Allergic Rhinitis
- Bentrio® meets primary efficacy endpoint in NASAR clinical trial in seasonal allergic rhinitis
- Clinically relevant and statistically significant improvement in Total Nasal Symptom Score over saline nasal spray control (p = 0.012)
- Bentrio efficacy and tolerability rated as "good" or "very good" by
63.5% and73.5% of study participants - NASAR concludes Altamira's clinical development program for Bentrio in allergic rhinitis
HAMILTON, BERMUDA / ACCESSWIRE / May 24, 2023 / Altamira Therapeutics Ltd. (Nasdaq:CYTO), a company dedicated to developing therapeutics that address important unmet medical needs, today announced positive and statistically significant top-line results from the randomized controlled NASAR clinical trial evaluating its Bentrio nasal spray in patients with seasonal allergic rhinitis (SAR). Bentrio nasal spray is formulated as a drug-free and preservative-free gel emulsion designed to help protect against airborne allergens such as pollen or house dust mites.
The NASAR trial enrolled 100 SAR patients in Australia who were randomized at a 1:1 ratio to receive either Bentrio or saline nasal spray for two weeks via self-administration three times per day, or as needed. For eligibility, patients had to have a baseline reflective Total Nasal Symptom Score (rTNSS) of at least 5 points out of 12, referring to the worst level of nasal congestion, sneezing, nasal itching, and rhinorrhea (runny nose) within the past 24 hours averaged over a one-week treatment-free run-in period. The primary efficacy endpoint was defined as the difference in the average rTNSS over the subsequent 2-week treatment period between Bentrio and saline nasal spray, the current standard of care in drug-free SAR management. The change in mean rTNSS over two weeks is generally accepted as a primary efficacy endpoint for SAR trials and is also recommended by the FDA.
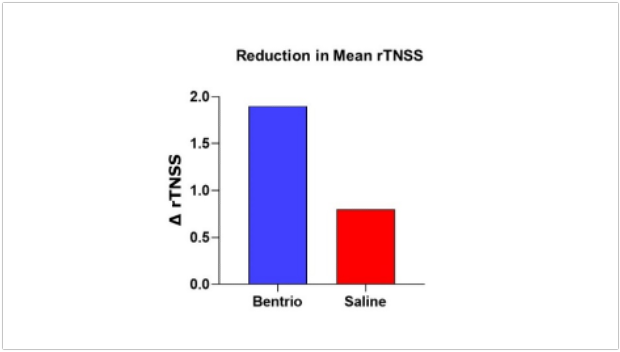
Graph above compares reduction in mean reflective Total Nasal Symptom Score (rTNSS) with Bentrio vs. saline nasal spray control over two weeks of treatment from baseline
The rTNSS decreased in the Bentrio group from 6.9 points in the pre-treatment period to an average of 5.0 points over the 14-day treatment period (i.e. -1.9 points), while the saline spray group showed a decrease from 6.9 to 6.2 points (i.e. -0.8 points) (see ‘Mean rTNSS' graph). The reduction in nasal symptoms conferred by Bentrio was thus 2.5 times larger than with saline nasal spray. The difference in rTNSS reduction of 1.1 points in favor of Bentrio was statistically significant in the ANCOVA model (LSmeans; p = 0.012;

Graph above compares patient rating of treatment efficacy with Bentrio vs. saline nasal spray control after two weeks of treatment
The treatment effect shown with Bentrio was well above the minimal clinically important difference of 0.28 points.
"We are thrilled with the strong outcomes from the NASAR trial, which we obtained under real-life conditions during the allergy season in Australia," commented Thomas Meyer, Altamira Therapeutics' founder, Chairman, and CEO. "The large magnitude of the reduction of nasal symptoms demonstrates that effective protection against seasonal allergic rhinitis is very well possible with a drug-free treatment. There are many patients who do not respond well to medicated nasal sprays either for lack of efficacy or due to tolerability issues with preservatives or other ingredients. I would like to extend my sincere appreciation to all the patients and investigators involved in the NASAR trial who helped us to reach this important clinical milestone.
"Further, the NASAR results show for Bentrio a statistically significant and clinically highly relevant improvement in efficacy over the current standard of care saline nasal spray treatment," Mr. Meyer added. "Whereas a saline nasal spray aims to rinse out allergen particles, Bentrio forms a thin protective layer which prevents contact of those particles with the nasal mucosa and helps to remove them through natural mucociliary clearance. As already demonstrated in a previous trial, Bentrio stays for about 3.5 hours within the nasal cavity where it can exert its protective effects. In contrast, saline spray is present for only about one hour and provides narrower distribution and less coverage within the nasal cavity.
"With the completion of the NASAR trial, Altamira has concluded its clinical development of Bentrio in allergic rhinitis management. In 2023, the US sales of over-the-counter allergy remedies are estimated to reach close to
About Bentrio
Bentrio is an OTC drug-free nasal spray for personal protection against airborne allergens and, where approved, against airborne viruses. Upon application into the nose, Bentrio forms a protective gel layer on the nasal mucosa. This thin film is designed to prevent the contact of allergens (or virus particles) with cells; in addition, the composition serves to bind such particles and help with their discharge. Together, this is designed to promote alleviation of allergic symptoms (or mitigate upper respiratory tract viral infections). For more info, visit: https://www.aurismedical.com/legacy-programs/bentrio
About Altamira Therapeutics
Altamira Therapeutics (Nasdaq:CYTO) is dedicated to developing and commercializing RNA delivery technology for extrahepatic targets (OligoPhore™ / SemaPhore™ platforms). The Company currently has two flagship siRNA programs in preclinical development beyond in vivo proof of concept: AM-401 for KRAS driven cancer and AM-411 for rheumatoid arthritis. The versatile delivery platform is also suited for mRNA and other types of RNA therapeutics and is planned to be leveraged via out-licensing to pharma or biotech companies. In addition, Altamira is in the process of divesting and/or out-licensing its legacy assets in allergology and viral infection (Bentrio® OTC nasal spray; commercial) and inner ear therapeutics (AM-125 nasal spray for vertigo; post Phase 2; Keyzilen® and Sonsuvi® for tinnitus and hearing loss; Phase 3). Founded in 2003, Altamira is headquartered in Hamilton, Bermuda, with its main operations in Basel, Switzerland. For more information, visit: https://altamiratherapeutics.com/
Forward-Looking Statements
This press release may contain statements that constitute "forward-looking statements" within the meaning of Section 27A of the Securities Act of 1933 and Section 21E of the Securities Exchange Act of 1934. Forward-looking statements are statements other than historical facts and may include statements that address future operating, financial or business performance or Altamira Therapeutics' strategies or expectations. In some cases, you can identify these statements by forward-looking words such as "may", "might", "will", "should", "expects", "plans", "anticipates", "believes", "estimates", "predicts", "projects", "potential", "outlook" or "continue", or the negative of these terms or other comparable terminology. Forward-looking statements are based on management's current expectations and beliefs and involve significant risks and uncertainties that could cause actual results, developments and business decisions to differ materially from those contemplated by these statements. These risks and uncertainties include, but are not limited to, the success of the continued commercialization of Bentrio and success of strategic transactions, including licensing or partnering, with respect to Bentrio or any other legacy assets, Altamira Therapeutics' need for and ability to raise substantial additional funding to continue the development of its product candidates, the timing and conduct of clinical trials of Altamira Therapeutics' product candidates, the clinical utility of Altamira Therapeutics' product candidates, the timing or likelihood of regulatory filings and approvals, Altamira Therapeutics' intellectual property position and Altamira Therapeutics' financial position, including the impact of any future acquisitions, dispositions, partnerships, license transactions or changes to Altamira Therapeutics' capital structure, including future securities offerings. These risks and uncertainties also include, but are not limited to, those described under the caption "Risk Factors" in Altamira Therapeutics' Annual Report on Form 20-F for the year ended December 31, 2022, and in Altamira Therapeutics' other filings with the SEC, which are available free of charge on the Securities Exchange Commission's website at: www.sec.gov. Should one or more of these risks or uncertainties materialize, or should underlying assumptions prove incorrect, actual results may vary materially from those indicated. All forward-looking statements and all subsequent written and oral forward-looking statements attributable to Altamira Therapeutics or to persons acting on behalf of Altamira Therapeutics are expressly qualified in their entirety by reference to these risks and uncertainties. You should not place undue reliance on forward-looking statements. Forward-looking statements speak only as of the date they are made, and Altamira Therapeutics does not undertake any obligation to update them in light of new information, future developments or otherwise, except as may be required under applicable law.
CONTACT:
Hear@altamiratherapeutics.com
800-460-0183
SOURCE: Altamira Therapeutics Ltd.
View source version on accesswire.com:
https://www.accesswire.com/756785/Altamira-Therapeutics-Reports-Positive-Top-Line-Data-from-Bentrio-Clinical-Trial-in-Seasonal-Allergic-Rhinitis







