Protagonist Reports Fourth Quarter and Full Year 2025 Financial Results and Provides Corporate Update
Rhea-AI Summary
Protagonist (Nasdaq:PTGX) reported Q4 and full-year 2025 results and a corporate update on Feb 25, 2026. Cash and marketable securities were $646.0M at year-end. The company submitted an NDA for rusfertide, expects a U.S. opt-out decision window in Q2 2026, and anticipates a 2026 U.S. decision and potential launch for ICOTYDE.
License and collaboration revenue fell to $46.0M for 2025 from $434.4M in 2024; net loss was $130.1M for 2025. PN-881 Phase 1 is expected complete by mid-2026.
Positive
- Cash and marketable securities of $646.0M at Dec 31, 2025
- NDA submitted for rusfertide with potential 2026 approval and launch
- U.S. regulatory decision for ICOTYDE anticipated in 2026 with possible launch
- PN-881 Phase 1 expected complete by mid-2026, advancing pipeline
Negative
- License and collaboration revenue declined to $46.0M in 2025 from $434.4M in 2024
- Net loss of $130.1M for full year 2025 versus net income in 2024
- Q4 2025 license revenue dropped to $7.4M from $170.6M in Q4 2024
Key Figures
Market Reality Check
Peers on Argus
PTGX gained 0.92% with mixed peer moves: MIRM, AAPG, AKRO modestly positive (up to 0.7%), while MTSR and ZLAB were slightly negative, supporting a company-specific reaction.
Previous Earnings Reports
| Date | Event | Sentiment | Move | Catalyst |
|---|---|---|---|---|
| Nov 06 | Q3 2025 earnings | Neutral | -0.3% | Q3 2025 results with NDA progress and strong cash runway to 2028. |
| Aug 06 | Q2 2025 earnings | Negative | -5.4% | Q2 loss and modest revenue alongside pipeline advances and obesity assets. |
| May 06 | Q1 2025 earnings | Positive | +2.6% | Positive Phase 3 results for rusfertide and icotrokinra with strong cash. |
| Feb 21 | FY 2024 earnings | Positive | +0.7% | Large 2024 revenue and net income from Takeda and JNJ milestones. |
| Nov 07 | Q3 2024 earnings | Negative | -2.9% | Q3 2024 loss despite substantial collaboration revenue and trial milestones. |
Earnings releases have produced an average -1.07% move, with a mix of positive and negative reactions and no consistent directional pattern.
Over the last five earnings cycles from Nov 2024 through Nov 2025, Protagonist transitioned from milestone-driven profitability in 2024 to loss-making but well-funded development in 2025. Prior updates highlighted rising cash balances (up to $697.9M in Q1 2025), major milestones such as the $165.0M icotrokinra payment, and progression of rusfertide and icotrokinra through Phase 3 and NDA submissions. Today’s full-year 2025 results extend that narrative, emphasizing regulatory filings, pipeline expansion, and a shift from milestone revenue to R&D investment.
Historical Comparison
Past five earnings releases saw an average move of -1.07%. Today’s modest 0.92% gain after 2025 results is slightly more positive than typical earnings reactions.
Earnings updates progressed from milestone-driven profitability in 2024 to 2025 results focused on NDA submissions for icotrokinra and rusfertide, growing cash balances, and expansion into obesity and oral IL-17/hepcidin programs.
Regulatory & Risk Context
The company has an effective S-3ASR shelf registration filed on 2025-08-06 with no recorded usage to date (usage count 0). This provides pre-cleared flexibility to issue securities if the company chooses, though no specific amounts or transactions are indicated in the current data.
Market Pulse Summary
This announcement combines 2025 earnings with clear milestones: NDAs for rusfertide and ICOTYDE, Phase 3 progress, and new obesity and hepcidin programs, all supported by $646.0 million in year-end cash. Revenue fell sharply versus the $434.4 million booked in 2024, reflecting fewer milestones and a $130.1 million net loss. Investors may track upcoming U.S. regulatory decisions, PN-881 Phase 1 completion by mid-2026, and any use of the effective S-3ASR shelf when assessing future dilution and growth potential.
Key Terms
new drug application regulatory
nda regulatory
phase 3 medical
randomized medical
double-blind medical
placebo-controlled medical
AI-generated analysis. Not financial advice.
NDA for rusfertide submitted to the US Food and Drug Administration (FDA), with potential approval and launch this year
Company expects to opt-out of the 50:50 profit and loss sharing arrangement for rusfertide with Takeda during a 90-day window expected to open in Q2
U.S. regulatory decision for ICOTYDETM (icotrokinra) anticipated in 2026 with
potential launch this year
PN-881 Phase 1 completion expected by mid-2026
NEWARK, CALIFORNIA / ACCESS Newswire / February 25, 2026 / Protagonist Therapeutics (Nasdaq:PTGX) ("Protagonist" or "the Company") today reported financial results for the fourth quarter and full year ended December 31, 2025 and provided a corporate update.
"In 2025, Protagonist reached new heights with multiple successful Phase 3 outcomes and two NDA filings of our partnered assets, ICOTYDE and rusfertide," said Dinesh V. Patel, Ph.D., the Company's President and CEO. "We see the next 12 to 24 months as a period of significant growth and value creation for Protagonist, driven by a combination of the anticipated regulatory and commercial milestones and royalties from ICOTYDE and rusfertide and the continued advancement of our robust R&D pipeline comprised of the oral IL-17 peptide antagonist, our obesity dual and triple agonists, and our oral hepcidin functional mimetic. We are well equipped to fund all our internal wholly owned programs to clinical proof-of-concept with the cash on hand and potential revenue from the partnered assets."
Fourth Quarter 2025 Recent Developments and Upcoming Milestones
Rusfertide
Under the terms of its License and Collaboration Agreement with Takeda Pharmaceuticals USA, Inc., Protagonist has the right to opt out of the 50:50 profit and loss sharing arrangement in the U.S. during the 90-day period beginning 120 days after filing of a New Drug Application with the FDA for Rusfertide for polycythemia vera. We currently expect to exercise that right in the second quarter of 2026.
ICOTYDETM (Icotrokinra)
A U.S. regulatory decision is anticipated in 2026, followed by commercial launch this year, if FDA approval is granted.
Primary endpoint enrollment completion is expected in 2026 for:
The Phase 3 ICONIC-ASCEND multicenter, randomized, double-blind, placebo-controlled, and ustekinumab active comparator-controlled study to evaluate the efficacy and safety of icotrokinra for the treatment of participants with moderate to severe plaque psoriasis (NCT06934226).
The Phase 3, multicenter, randomized, double-blind, placebo-controlled study evaluating the efficacy and safety of icotrokinra for the treatment of biologic-naïve participants with active psoriatic arthritis (NCT06878404).
Clinical Programs
The Company expects its Phase 1 study of PN-881 to be complete by mid-2026, informing subsequent clinical development plans.
Discovery Programs
Recently, Protagonist announced two new wholly owned development candidates:
PN-458, a novel dual GLP-GIP agonist for obesity, and
PN-8047, an oral hepcidin functional mimetic complementing rusfertide, an injectable hepcidin mimetic.
Additionally, the Company added IL-4Rα and amylin as high-priority discovery programs to further expand and strengthen its pipeline.
Fourth Quarter and Full Year 2025 Financial Results
Cash, Cash Equivalents and Marketable Securities: Cash, cash equivalents, and marketable securities as of December 31, 2025, were
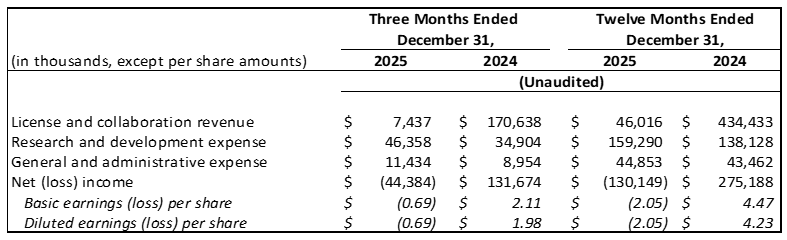
Revenue: License and collaboration revenue is derived from the Company's License and Collaboration Agreement with JNJ, (the "JNJ Agreement"), and its License and Collaboration Agreement with Takeda (the "Takeda Agreement").
License and collaboration revenue decreased by
$163.2 million from$170.6 million for the fourth quarter of 2024 to$7.4 million for the fourth quarter of 2025. License and collaboration revenue decreased by$388.4 million from$434.4 million for the full year 2024 to$46.0 million for the full year 2025. The decrease in revenue was primarily attributable to lower milestone and collaboration revenue, which is highly variable and dependent upon factors such as the timing of when regulatory and sales milestones are achieved, if at all, and the accounting for any upfront payments associated with any existing or new agreements.
License and collaboration revenue of
$7.4 million for the fourth quarter of 2025 was comprised of development services we provided during the period under the Takeda Agreement. License and collaboration revenue of$170.6 million for the fourth quarter of 2024 included (i) achievement of a non-refundable$165.0 m ilestone under the JNJ Agreement, and (ii) development services we provided during the period under the Takeda Agreement.
License and collaboration revenue of
$46.0 million for the full year 2025 was comprised of (i) proportional recognition of a$25.0 million milestone earned from Takeda in Q1 25, and (ii) development services we provided during the period under the Takeda agreement. License and collaboration revenue of$434.4 million for the full year 2024 included (i)$254.1 million of the$300.0 million initial transaction price for the Takeda Agreement allocated to the rusfertide license upon effectiveness of the agreement, (ii) achievement of a non-refundable$165.0 m ilestone under the JNJ Agreement earned in Q4 24, and (iii) development services we provided during the period under the Takeda Agreement.
Research and Development Expenses: Increased by
General and Administrative Expenses: Increased by
Net (Loss) Income: Net loss was
About Protagonist
Protagonist Therapeutics is a discovery through late-stage development biopharmaceutical company. Two novel peptides derived from Protagonist's proprietary discovery platform are currently in advanced Phase 3 clinical development, with a New Drug Application (NDA) for ICOTYDETM (icotrokinra) under review at the FDA and an NDA for rusfertide submitted in December 2025. ICOTYDE is a first-in-class investigational targeted oral peptide that selectively blocks the Interleukin-23 receptor ("IL-23R"), which is licensed to Janssen Biotech, Inc., a Johnson & Johnson company. Following ICOTYDE's joint discovery by Protagonist and Johnson & Johnson scientists pursuant to the companies' IL-23R collaboration, Protagonist was primarily responsible for the development of ICOTYDE through Phase 1, with Johnson & Johnson assuming responsibility for development in Phase 2 and beyond. Rusfertide, a mimetic of the natural hormone hepcidin, is currently in development for the rare blood disorder polycythemia vera. Rusfertide is being co-developed and may be co-commercialized with Takeda Pharmaceuticals pursuant to a worldwide collaboration and license agreement under which the Company was primarily responsible for development through NDA filing. The Company also has a number of preclinical stage drug discovery programs addressing clinically and commercially validated targets, including an oral IL-17 peptide antagonist, obesity dual and triple agonists, an oral hepcidin functional mimetic, and the recently announced IL-4 and amylin programs.
More information on Protagonist, its pipeline drug candidates, and clinical studies can be found on the Company's website at https://www.protagonist-inc.com/.
Cautionary Note on Forward-Looking Statements
This press release contains forward-looking statements for purposes of the safe harbor provisions of the Private Securities Litigation Reform Act of 1995. Forward-looking statements include statements regarding potential timing of regulatory actions, clinical trial results, and potential revenue from the Company's collaborations with Takeda and Johnson & Johnson. In some cases, you can identify these statements by forward-looking words such as "anticipate," "believe," "may," "will," "expect," or the negative or plural of these words or similar expressions. Forward-looking statements are not guarantees of future performance and are subject to risks and uncertainties that could cause actual results and events to differ materially from those anticipated, including, but not limited to, our ability to develop and commercialize our product candidates, our ability to earn milestone payments under our collaboration agreements with Janssen and Takeda, our ability to use and expand our programs to build a pipeline of product candidates, our ability to obtain and maintain regulatory approval of our product candidates, our ability to operate in a competitive industry and compete successfully against competitors that have greater resources than we do, and our ability to obtain and adequately protect intellectual property rights for our product candidates. Additional information concerning these and other risk factors affecting our business can be found in our periodic filings with the Securities and Exchange Commission, including under the heading "Risk Factors" contained in our most recently filed periodic reports on Form 10-K and Form 10-Q filed with the Securities and Exchange Commission. Forward-looking statements are not guarantees of future performance, and our actual results of operations, financial condition, and liquidity, and the development of the industry in which we operate, may differ materially from the forward-looking statements contained in this press release. Any forward-looking statements that we make in this press release speak only as of the date of this press release. We assume no obligation to update our forward-looking statements, whether as a result of new information, future events, or otherwise, after the date of this press release.
Investor Relations Contact
Corey Davis, Ph.D.
LifeSci Advisors
cdavis@lifesciadvisors.com
+1 212 915 2577
Media Relations Contact
Virginia Amann
ENTENTE Network of Companies
virginiaamann@ententeinc.com
+1 833 500 0061 ext 1
PROTAGONIST THERAPEUTICS, INC.
Consolidated Statements of Operations
(Amounts in thousands except share and per share data)
Three Months Ended | Twelve Months Ended | |||||||||||||||
December 31, | December 31, | |||||||||||||||
2025 | 2024 | 2025 | 2024 | |||||||||||||
License and collaboration revenue | $ | 7,437 | $ | 170,638 | $ | 46,016 | $ | 434,433 | ||||||||
Operating expense: | ||||||||||||||||
Research and development (1) | 46,358 | 34,904 | 159,290 | 138,128 | ||||||||||||
General and administrative (1) | 11,434 | 8,954 | 44,853 | 43,462 | ||||||||||||
Total operating expense | 57,792 | 43,858 | 204,143 | 181,590 | ||||||||||||
Loss (income) from operations | (50,355 | ) | 126,780 | (158,127 | ) | 252,843 | ||||||||||
Interest income | 6,761 | 6,853 | 28,789 | 26,315 | ||||||||||||
Other (expense) income, net | (124 | ) | 31 | 27 | 250 | |||||||||||
(Loss) income before income tax expense | (43,718 | ) | 133,664 | (129,311 | ) | 279,408 | ||||||||||
Income tax expense | 666 | 1,990 | 838 | 4,220 | ||||||||||||
Net (loss) income | $ | (44,384 | ) | $ | 131,674 | $ | (130,149 | ) | $ | 275,188 | ||||||
Net (loss) income per share, basic | $ | (0.69 | ) | $ | 2.11 | $ | (2.05 | ) | $ | 4.47 | ||||||
Net (loss) income per share, diluted | $ | (0.69 | ) | $ | 1.98 | $ | (2.05 | ) | $ | 4.23 | ||||||
Weighted-average shares used to compute net (loss) income per share, basic | 64,031,592 | 62,328,468 | 63,573,048 | 61,566,989 | ||||||||||||
Weighted-average shares used to compute net (loss) income per share, diluted | 64,031,592 | 66,406,817 | 63,573,048 | 65,077,722 | ||||||||||||
(1) Amount includes non-cash stock-based compensation expense.
Stock-based Compensation
(In thousands)
Three Months Ended | Twelve Months Ended | |||||||||||||||
December 31, | December 31, | |||||||||||||||
2025 | 2024 | 2025 | 2024 | |||||||||||||
Research and development | $ | 6,121 | $ | 5,322 | $ | 26,422 | $ | 20,919 | ||||||||
General and administrative | 4,580 | 3,771 | 19,552 | 16,635 | ||||||||||||
Total stock-based compensation expense | $ | 10,701 | $ | 9,093 | $ | 45,974 | $ | 37,554 | ||||||||
PROTAGONIST THERAPEUTICS, INC.
Selected Consolidated Balance Sheet Data
(In thousands)
December 31, | December 31, | |||||||
2025 | 2024 | |||||||
Cash, cash equivalents and marketable securities | $ | 646,002 | $ | 559,165 | ||||
Working capital | 532,133 | 544,243 | ||||||
Total assets | 668,188 | 744,725 | ||||||
Deferred revenue | 9,550 | 30,567 | ||||||
Accumulated deficit | (470,671 | ) | (340,522 | ) | ||||
Total stockholders' equity | 614,707 | 675,295 | ||||||
SOURCE: Protagonist Therapeutics
View the original press release on ACCESS Newswire







