PolyPid to Present Data from SHIELD II Phase 3 Trial Demonstrating Reduced Severity of Surgical Site Infection at Surgical Infection Society 2026 Annual Meeting
Rhea-AI Summary
PolyPid (Nasdaq: PYPD) announced presentation of Phase 3 SHIELD II ASEPSIS score analysis showing a 64% relative risk reduction (p=0.0103) in patients with ASEPSIS>20 and a previously reported 60% RRR in SSI incidence (p=0.0013) after abdominal colorectal surgery. D-PLEX100 holds FDA Breakthrough Therapy designation for SSI prevention.
The data suggest fewer high-severity wound infections and potential reductions in antibiotic use, hospital stay, and resource utilization; results will be presented May 6, 2026 at the Surgical Infection Society annual meeting.
Positive
- ASEPSIS >20 reduced by 64% RRR (p=0.0103)
- SSI incidence reduced by 60% RRR (p=0.0013)
- D-PLEX100 holds FDA Breakthrough Therapy designation
Negative
- ASEPSIS was a secondary endpoint, not the primary trial endpoint
- Findings presented at a conference; full peer-reviewed dataset not yet published
News Market Reaction – PYPD
On the day this news was published, PYPD declined 3.28%, reflecting a moderate negative market reaction. This price movement removed approximately $3M from the company's valuation, bringing the market cap to $84.54M at that time.
Data tracked by StockTitan Argus on the day of publication.
Key Figures
Market Reality Check
Peers on Argus
PYPD slipped 0.87% while key biotech peers were mixed: CUE up 2.57%, CAMP up 0.45%, KPTI and VTVT down, PDSB flat. Momentum scanner only flagged one peer (SER) moving up, supporting a stock-specific read on this clinical update.
Previous Clinical trial Reports
| Date | Event | Sentiment | Move | Catalyst |
|---|---|---|---|---|
| Apr 13 | PK data presentation | Positive | +2.1% | Showed 32-day local doxycycline release with minimal systemic exposure. |
| Mar 31 | NDA initiation | Positive | +4.0% | Started rolling NDA submission for D-PLEX100 under Fast Track status. |
| Mar 17 | PDUFA fee waiver | Positive | +3.6% | Received $4.3M PDUFA fee waiver and reiterated 60% infection risk reduction. |
| Dec 03 | Pre-NDA meeting | Positive | +4.8% | FDA pre‑NDA minutes supported rolling NDA based on SHIELD II package. |
| Jun 16 | Warrant financing | Positive | -3.8% | Raised $26.7M via warrant exercise following positive SHIELD II data. |
Recent clinical-trial and regulatory milestones for D-PLEX100 have generally been followed by modest positive moves, with one funding-related divergence.
Over the past year, PolyPid has advanced D-PLEX100 through key Phase 3 SHIELD II and regulatory milestones. Positive FDA pre‑NDA feedback in Dec 2025 supported a rolling NDA, followed by a $4.3M PDUFA fee waiver and warrant-funded $26.7M capital raise tied to successful SHIELD II results. In Mar–Apr 2026, the company began its rolling NDA and highlighted sustained local doxycycline release data. Today’s SHIELD II ASEPSIS severity analysis extends that clinical narrative with more granular outcome data.
Historical Comparison
Past clinical-trial headlines for PYPD moved an average of 2.14%, suggesting data-driven updates like this SHIELD II ASEPSIS analysis have historically prompted moderate but generally positive reactions.
Clinical-trial news shows a progression from Phase 3 SHIELD II success to supportive FDA pre‑NDA feedback, a PDUFA fee waiver, rolling NDA initiation, and multiple detailed data presentations, building a consistent D-PLEX100 narrative.
Market Pulse Summary
This announcement details additional Phase 3 SHIELD II outcomes, showing statistically significant 64% and 60% relative risk reductions in severe wound infections and SSI incidence with D-PLEX100. It complements prior data on sustained 30‑day local doxycycline release and supports the ongoing NDA process. Historically, similar clinical-trial updates have produced modestly positive moves. Investors may watch for further FDA interactions, partnership progress, and any new data on safety, durability of effect, and hospital resource utilization.
Key Terms
asepsis score medical
surgical site infections medical
broad-spectrum antibiotic medical
doxycycline medical
phase 3 medical
breakthrough therapy designation regulatory
AI-generated analysis. Not financial advice.
Analysis demonstrates statistically significant reduction in the severity of surgical wound infections, with potential to lower hospital resource utilization following abdominal colorectal surgery
PETACH TIKVA, Israel, April 28, 2026 (GLOBE NEWSWIRE) -- PolyPid Ltd. (Nasdaq: PYPD) (“PolyPid” or the “Company”), an innovative biopharmaceutical company dedicated to improving patient outcomes by elevating treatment effectiveness, right where care begins, today announced that it will present at the 45th Annual Meeting of the Surgical Infection Society, taking place on May 5-6, 2026, at the Coronado Island Marriott in Coronado, California. The presentation will focus on a detailed analysis of the ASEPSIS (Additional treatment, Serous discharge, Erythema, Purulent exudate, Separation of deep tissues, Isolation of bacteria, and prolonged Stay as inpatient) score data from the Company's Phase 3 SHIELD II trial of D-PLEX100.
The presentation will be delivered by Dr. Robert G. Sawyer, MD, FSIS, FACS, FCCM, FIDSA, a Professor and Chair of the Department of Surgery at Western Michigan University Homer Stryker M.D. School of Medicine. Dr. Sawyer also serves as Senior Associate Dean for Research and Editor-in-Chief of the journal Surgical Infections.
The ASEPSIS score is a validated, objective clinical scoring system used to assess the severity of surgical wound infections. It integrates parameters such as wound appearance, antibiotic use, drainage of pus, isolation of pathogenic bacteria, and length of hospital stay. As previously reported in the SHIELD II topline results, treatment with D-PLEX100 together with the standard of care resulted in a
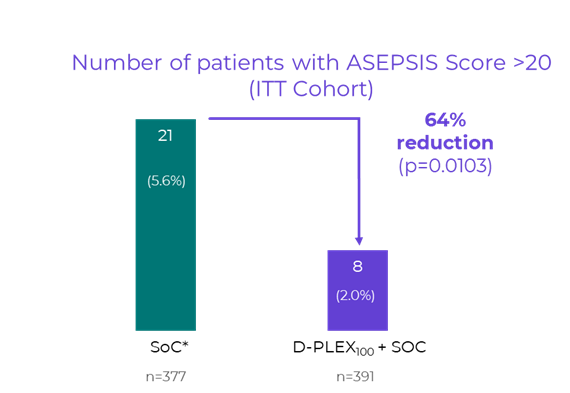
"The ASEPSIS score outcomes demonstrate not only that D-PLEX₁₀₀ reduces surgical site infections (“SSI”) events by
Surgical Infection Society 2026 Annual Meeting Presentation Details
| Title: | Evaluation of ASEPSIS Score as a Secondary Endpoint in the Phase 3 SHIELD II Trial of D-PLEX100 in Colorectal Surgery |
| Presenter: | Robert G. Sawyer, MD, FSIS, FACS, FCCM, FIDSA, Professor and Chair, Department of Surgery, Western Michigan University Homer Stryker M.D. School of Medicine; Senior Associate Dean for Research; Editor-in-Chief, Surgical Infections |
| Session: | Scientific Session III |
| Date/Time: | Wednesday, May 6, 2026 | 8:00 a.m. – 9:00 a.m. PT |
| Location: | Coronado Island Marriott, Coronado, California |
About D-PLEX100
D-PLEX100, PolyPid’s lead product candidate, is designed to provide local prolonged and controlled anti-bacterial activity directly at the surgical site to prevent surgical site infections (“SSIs”). Following the administration of D-PLEX100 into the surgical site, PolyPid’s delivery technology, Kynatrix, pairs with Active Pharmaceutical Ingredients, enabling a prolonged and continuous release of the broad-spectrum antibiotic doxycycline, resulting in a high local concentration of the drug for a period of 30 days for the prevention of SSIs, with additional potential to prevent SSIs caused by antibiotic-resistant bacteria at the surgical site. D-PLEX100 recently demonstrated positive results in the Phase 3 SHIELD II trial, achieving a statistically significant
About PolyPid
PolyPid Ltd. (Nasdaq: PYPD) is an innovative biopharmaceutical company dedicated to elevating treatment effectiveness, right where care begins. The Company develops long-acting, controlled-release drugs designed to deliver therapy precisely at the site of care, addressing critical unmet medical needs across a wide and diverse pipeline spanning surgical care, metabolic diseases, and beyond. PolyPid’s lead product, D-PLEX100, successfully met its primary and all key secondary endpoints in the landmark Phase 3 SHIELD II trial for the prevention of surgical site infections. Guided by a commitment to precision and innovation, PolyPid is redefining how therapies perform and raise the standard of patient care. For additional Company information, please visit http://www.polypid.com and follow us on Twitter (X) and LinkedIn.
Forward-looking Statements
This press release contains “forward-looking statements” within the meaning of the Private Securities Litigation Reform Act and other securities laws. Words such as “expects,” “anticipates,” “intends,” “plans,” “believes,” “seeks,” “estimates” and similar expressions or variations of such words are intended to identify forward-looking statements. For example, the Company is using forward-looking statements when it discusses the potential clinical and economic benefits of D-PLEX100, including its potential to reduce the incidence of severe wound infections, decrease wound complications and the use of intravenous antibiotics, enable earlier hospital discharge, and lower hospital resource utilization, and the potential role of D-PLEX100 in improving the prevention of SSIs. Forward-looking statements are not historical facts, and are based upon management’s current expectations, beliefs and projections, many of which, by their nature, are inherently uncertain. Such expectations, beliefs and projections are expressed in good faith. However, there can be no assurance that management’s expectations, beliefs and projections will be achieved, and actual results may differ materially from what is expressed in or indicated by the forward-looking statements. Forward-looking statements are subject to risks and uncertainties that could cause actual performance or results to differ materially from those expressed in the forward-looking statements. For a more detailed description of the risks and uncertainties affecting the Company, reference is made to the Company’s reports filed from time to time with the Securities and Exchange Commission, including, but not limited to, the risks detailed in the Company’s Annual Report on Form 20-F filed on February 25, 2026. Forward-looking statements speak only as of the date the statements are made. The Company assumes no obligation to update forward-looking statements to reflect actual results, subsequent events or circumstances, changes in assumptions or changes in other factors affecting forward-looking information except to the extent required by applicable securities laws. If the Company does update one or more forward-looking statements, no inference should be drawn that the Company will make additional updates with respect thereto or with respect to other forward-looking statements. References and links to websites have been provided as a convenience, and the information contained on such websites is not incorporated by reference into this press release. PolyPid is not responsible for the contents of third-party websites.
Company Contact:
PolyPid Ltd.
Ori Warshavsky
908-858-5995
IR@Polypid.com
Investor Relations Contact:
Arx Investor Relations
North American Equities Desk
polypid@arxhq.com
A photo accompanying this announcement is available at https://www.globenewswire.com/NewsRoom/AttachmentNg/43a11cdb-f86b-4d84-bfb0-1870a0a2cb4e

