Recent Study Confirms Fundamental Correlation Between Severity of Obstructive Sleep Apnea and Oral Anatomy
Rhea-AI Summary
Vivos Therapeutics (NASDAQ: VVOS) highlighted a peer-reviewed study linking narrower maxillary intermolar distance and higher palatal height to greater obstructive sleep apnea (OSA) severity, reporting a strong association (p<0.001) across 100 patients.
The company noted its FDA 510(k) clearance in November 2024 for Vivos CARE devices to treat severe OSA, U.S. clearance for adults and children, and asserted many patients finish treatment in under 12 months, often reducing the need for nightly CPAP or other lifetime interventions.
AI-generated analysis. Not financial advice.
Positive
- FDA 510(k) clearance granted in November 2024 to treat severe OSA
- Study association between IMD/PH and AHI with statistical significance (p<0.001)
- Clinical reach—Vivos-trained providers treated tens of thousands of OSA patients
- Short treatment claim: many patients finish therapy in less than 12 months
Negative
- Study sample size was 100 patients, limiting broad generalizability
- Correlation study links anatomy to OSA severity but does not alone prove long-term treatment outcomes
News Market Reaction – VVOS
On the day this news was published, VVOS gained 1.54%, reflecting a mild positive market reaction. Argus tracked a peak move of +5.1% during that session. Our momentum scanner triggered 3 alerts that day, indicating moderate trading interest and price volatility. This price movement added approximately $229K to the company's valuation, bringing the market cap to $15.07M at that time.
Data tracked by StockTitan Argus on the day of publication.
Key Figures
Market Reality Check
Peers on Argus
VVOS gained 4.84% with relatively light volume while only one peer (MYO) appeared on the momentum scanner, up 3.43% without same-day news, indicating today’s move looks more company-specific than sector-driven.
Historical Context
| Date | Event | Sentiment | Move | Catalyst |
|---|---|---|---|---|
| Feb 05 | Technology collaboration | Positive | +0.0% | Collaboration with SoundHealth to distribute FDA-cleared sleep and allergy devices. |
| Jan 20 | Warrant exercise close | Negative | -7.1% | Closing of warrant exercises raising $4.64M gross proceeds at reduced prices. |
| Jan 16 | Warrant exercise deal | Negative | -3.3% | Agreement for immediate warrant exercise and issuance of new private warrants. |
| Dec 16 | Sleep center opening | Positive | +0.0% | Opening of affiliated sleep testing and treatment center in Auburn Hills, Michigan. |
| Dec 15 | Investor fireside chat | Neutral | +0.0% | Announcement of CEO/CFO participation in a Water Tower Research fireside chat. |
Financing-related announcements have seen negative price reactions, while operational and partnership news often showed little immediate price response.
Over the past few months, Vivos reported collaborations, financing actions, and network expansion. A February 2026 collaboration with SoundHealth leveraged its network of over 2,000 providers but saw no notable price move. January warrant exercises generated roughly $4.64M in proceeds yet coincided with share price declines. Late 2025 brought expansion via a new sleep center near Detroit and an investor fireside chat, both with flat reactions. Today’s study-driven validation of Vivos CARE devices adds scientific support alongside that commercial and financing backdrop.
Regulatory & Risk Context
An effective S-3 shelf registration filed on 2026-02-13 covers 3,964,712 warrant shares at $2.09. The company would not receive proceeds from resales but could receive up to $8.29 million if all covered warrants are exercised for cash.
Market Pulse Summary
This announcement highlights peer-reviewed evidence linking craniofacial anatomy to OSA severity and reinforces FDA-cleared Vivos CARE devices, including a unique 510(k) clearance from November 2024 for severe OSA. The study’s 100-patient cohort and strong significance (p<0.001) support the airway-remodeling mechanism. In context of prior collaborations and financing moves, key items to watch include additional clinical publications, adoption across the 2,000+ provider network, and how capital-structure tools such as outstanding warrants interact with growth plans.
Key Terms
obstructive sleep apnea medical
apnea-hypopnea index medical
cpap medical
neurostimulation implants medical
510(k) clearance regulatory
mandibular advancement oral appliances medical
hypoxia medical
AI-generated analysis. Not financial advice.
Vivos CARE Oral Medical Devices Proven to Enhance Airways by Redeveloping Dental Arch Forms and Jaws
LITTLETON, Colo., March 17, 2026 (GLOBE NEWSWIRE) -- Vivos Therapeutics, Inc. (“Vivos” or the “Company’’) (NASDAQ: VVOS), a leading medical device and healthcare services company focused on the treatment of breathing related sleep disorders and associated chronic health conditions, today announced that an a recent study published in the Journal of Clinical Medicine supports Vivos’ long maintained belief that dental and facial morphology characterized by narrow palatal (upper) dental arches, high peaked palates, and recessed lower jaws were directly correlated to the prevalence and severity of obstructive sleep apnea (OSA) in adults and children.
Over the past decade, Vivos-trained providers have successfully treated tens of thousands of OSA patients using Vivos CARE devices to redevelop and enhance airways. Vivos CARE devices have been cleared by the U.S. Food and Drug Administration (FDA) for adult patients diagnosed with mild, moderate and severe OSA and moderate-to-severe OSA in children ages 6 to 17. Many Vivos patients finish treatment in less than 12 months with little or no remaining OSA symptoms. Thus, in the majority of such Vivos patients, there is no need for nightly lifetime intervention with CPAP, traditional oral appliances, or neurostimulation implants.
The recent study, the “Correlation Between Severity of Obstructive Sleep Apnea and Dental Arch Form in Adults,” first published in October 2025, affirms what the over 2,000 Vivos-trained providers see every day in clinical practice—that very small changes in the size, shape, and position of the dentition and jaws evoke significant changes in the form, function and patency of the human airway. The study conclusions indicate that maxillary morphology is associated with OSA severity, narrower intermolar distance (IMD) and greater palatal height (PH) linked to higher Apnea-Hypopnea Index (AHI). Larger airways are less prone to collapse (apnea / hypopnea events) during sleep. Fewer apnea / hypopnea events often means less sleep disruption, less blood oxygen desaturation (hypoxia) and better overall sleep architecture.
One hundred patients participated in the study, with 50 OSA patients and 50 controls. The OSA group exhibited significantly narrower transverse intermolar distance and higher palatal height compared to the control group. Linear regression analysis demonstrated a strong association (p<0.001) between these two key craniofacial parameters and OSA severity, as measured by the industry standard measure AHI. These results held true across all demographics. The figure below reflects the strong anatomical correlations in the study.
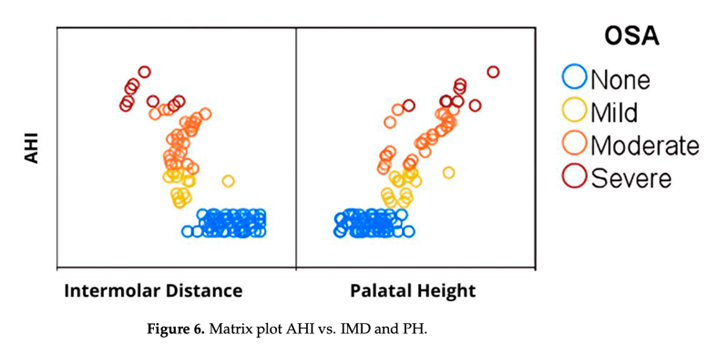
After years of research and product development, in November 2024 the FDA granted Vivos CARE devices an unprecedented 510(k) clearance to treat severe OSA patients. No other oral appliance had ever received such a clearance, nor has any similar clearance been granted since. Moreover, the mechanism of action in Vivos CARE devices is unique and capable of creating what are believed to be lasting results by gradually redeveloping and repositioning the dental arches and jaws.
Kirk Huntsman, CEO of Vivos Therapeutics, said, “This study reaffirms our longstanding view that cranial and oral morphology is directly correlated to not only the presence of OSA, but also its severity. Traditional treatments such as CPAP, mandibular advancement oral appliances, and neurostimulation implants, all act to temporarily manage symptoms, but require repeated nightly use over a lifetime to remain effective. Vivos CARE devices rehabilitate and restore optimal airway size and shape, allowing patients to breathe more fully and sleep more deeply. After nearly 50 years of limited and undesirable treatment options, patients now have a real choice that is both safe and effective.”
"This year, we estimate that several million Americans will undergo a sleep test. In our experience, nearly half of them will test positive for OSA. Those patients will need to make a serious and life-changing decision—what they will do about their serious medical condition. OSA doesn’t go away on its own, and it will likely get worse with time. If left untreated, it will affect their cardiovascular system, their neurocognitive abilities, their risk of diabetes, chronic fatigue, mental health and more. Yet over
About Vivos Therapeutics, Inc.
Vivos Therapeutics, Inc. (NASDAQ: VVOS) is a medical technology company focused on developing and commercializing innovative diagnostic and treatment methods for patients suffering from breathing and sleep issues arising from certain dentofacial abnormalities such as obstructive sleep apnea (OSA) and snoring in adults. Vivos’ devices have been cleared by the U.S. Food and Drug Administration (FDA) for adult patients diagnosed with all severity levels of OSA and moderate-to-severe OSA in children ages 6 to 17. Vivos’ groundbreaking Complete Airway Repositioning and Expansion (CARE) devices are the only FDA 510(k) cleared technology for treating severe OSA in adults and the first to receive clearance for treating moderate to severe OSA in children.
OSA affects over 1 billion people worldwide, yet
Founded in 2016 and based in Littleton, Colorado, Vivos is working to change this. Through innovative technology, education, and acquisitions of, or commercial collaborations with, sleep healthcare providers, Vivos is empowering healthcare providers to address the complex needs of OSA patients more thoroughly.
Vivos calls the use of its appliances and protocols to treat OSA The Vivos Method, which offers a proprietary, clinically effective solution that is nonsurgical, noninvasive, and nonpharmaceutical, providing hope to allow patients to Breathe New Life.
For more information, visit www.vivos.com.
Cautionary Note Regarding Forward-Looking Statements
This press release, including statements of the Company’s management and other parties made in connection therewith, contain “forward-looking statements” (as defined in Section 27A of the Securities Act of 1933, as amended, and Section 21E of the Securities Exchange Act of 1934, as amended) concerning future events. Words such as “may”, “would”, “should”, “expects”, “projects,” “potential,” “intends”, “plans”, “believes”, “anticipates”, “hopes”, “estimates”, “goal”. “aim” and variations of such words and similar expressions are intended to identify forward-looking statements. In this press release, forward-looking statements include, without limitation, those relating to the actual future impact on Vivos’ revenue and profitability from its operations or the benefits to Vivos of the study as described herein. These statements involve significant known and unknown risks and are based upon several assumptions and estimates, which are inherently subject to significant uncertainties and contingencies, many of which are beyond Vivos’ control. Actual results (including the actual results from the study and individual results of OSA treatment) may differ materially and adversely from those expressed or implied by such forward-looking statements. Factors that could cause actual results to differ materially include, but are not limited to: (i) the risk that Vivos may be unable to effectively market or continue to integrate business from the acquisition and alliance model into its own or otherwise implement sales, marketing and other strategies that increase revenues, (ii) the risk that some patients may not achieve the desired results from using Vivos’ products, (iii) risks associated with regulatory scrutiny of and adverse publicity in the sleep apnea diagnosis and treatment sector; (iv) the risk that Vivos may be unable to secure additional financing to continue operations, acquire additional sleep centers practices on reasonable terms, or maintain its Nasdaq listing when needed, if at all, (v) market and other conditions that could impact Vivos’ business or ability to obtain financing, and (vi) other risk factors described in Vivos’ filings with the Securities and Exchange Commission (“SEC”). Vivos’ filings can be obtained free of charge on the SEC’s website at www.sec.gov. Except to the extent required by law, Vivos expressly disclaims any obligations or undertaking to release publicly any updates or revisions to any forward-looking statements contained herein to reflect any change in Vivos’ expectations with respect thereto or any change in events, conditions, or circumstances on which any statement is based.
Media Inquiries:
Jennifer Hauser, Executive Assistant to the CEO
Investor Relations Contact
investors@vivoslife.com
A photo accompanying this announcement is available at https://www.globenewswire.com/NewsRoom/AttachmentNg/2bbc6139-7151-4edd-89dd-5253307b498b

