Artelo Biosciences Expansion into $16.3B Glaucoma Market, Via Fully Funded Study, Highlights Multi-Platform Pipeline Strength
Rhea-AI Summary
Artelo Biosciences (NASDAQ: ARTL) expands ART27.13 into the $16.3B glaucoma market via a fully funded investigator-sponsored trial while advancing three clinical programs in oncology support, neuropathic pain, and CNS disorders.
Key advantages: positive Phase 2 signals, MHRA guidance for ART12.11, FABP inhibitor Phase 1 safety, European patent protection to 2041, and a $25M equity facility.
Positive
- Investigator-sponsored glaucoma study funded externally targeting a $16.3B market
- ART27.13 Phase 2 interim weight signal (6.4% gain vs 5.4% placebo loss)
- ART26.12 completed Phase 1 with no serious adverse events and predictable PK
- ART12.11 received MHRA streamlined guidance and aligns with CBD research policy
- European patent protection for ART27.13 through 2041
Negative
- No announced pharma partnerships despite unsolicited expressions of interest
- Equity access facility ($25M expandable) may lead to dilution if drawn
- 1-for-3 reverse stock split could reduce trading liquidity for retail holders
- Investigator-sponsored trial outcomes are uncertain and not company-funded
Market Reaction – ARTL
Following this news, ARTL has declined 15.37%, reflecting a significant negative market reaction. Argus tracked a trough of -6.0% from its starting point during tracking. Our momentum scanner has triggered 39 alerts so far, indicating elevated trading interest and price volatility. The stock is currently trading at $4.90. This price movement has removed approximately $655K from the company's valuation.
Data tracked by StockTitan Argus (15 min delayed). Upgrade to Silver for real-time data.
Key Figures
Market Reality Check
Peers on Argus
ARTL fell 24.41% while only one scanned peer, HCWB, was also down (-3.2%). Other close peers showed mixed moves, pointing to a stock-specific reaction rather than a coordinated biotech-sector move.
Historical Context
| Date | Event | Sentiment | Move | Catalyst |
|---|---|---|---|---|
| Mar 18 | Glaucoma trial note | Positive | +50.7% | Vanderbilt analysis of fully funded ART27.13 glaucoma study and patent runway. |
| Mar 18 | Glaucoma study deal | Positive | +50.7% | Definitive IST agreement for ART27.13 in glaucoma funded by UK partners. |
| Mar 06 | Reverse stock split | Neutral | -2.6% | 3‑for‑1 reverse split to increase share price and adjust capital structure. |
| Feb 24 | Earnings and update | Neutral | +4.8% | Year-end results plus progress across ART27.13, ART26.12, and ART12.11 programs. |
| Dec 23 | CBD policy comment | Positive | -1.8% | Company response to CBD research executive order, highlighting ART12.11 profile. |
Positive glaucoma-related news on Mar 18 saw strong gains (~50.72%), while earlier positive CBD policy news in Dec 2025 coincided with a mild decline, showing occasional divergence on good news.
Over the past six months, ARTL has reported multiple clinical and corporate milestones. Two Mar 18 glaucoma updates tied to a fully funded investigator-sponsored trial drove a strong +50.72% reaction. A 3‑for‑1 reverse split announced on Mar 6 had only a modest impact. The Feb 24 business update combined positive clinical signals with a sizeable net loss, yet still saw a modest gain. Earlier, a Dec 23, 2025 CBD policy catalyst aligned with slightly negative trading, illustrating that not all positive disclosures translated into upside.
Market Pulse Summary
The stock is dropping -15.4% following this news. A negative reaction despite the bullish multi-asset narrative fits a pattern where strong stories have sometimes met skepticism, as seen when positive CBD policy news in Dec 2025 coincided with a -1.84% move. The current -24.41% decline came while the stock traded 93.25% below its 52-week high and below the 200-day MA, against a backdrop of prior going-concern language and frequent capital raises, factors that could have amplified downside pressure.
Key Terms
endocannabinoid system medical
cb2 receptor medical
pharmacokinetics medical
cocrystal medical
mhra regulatory
AI-generated analysis. Not financial advice.
Three clinical programs, one core molecule, and a capital-efficient strategy position Artelo Biosciences for sustained growth beyond a single indication
BRISTOL, Tenn., March 25, 2026 (GLOBE NEWSWIRE) -- Artelo Biosciences, Inc. (NASDAQ: ARTL) is drawing increased attention following its expansion into the
Artelo Biosciences is running three distinct clinical-stage programs — in oncology support, neuropathic pain, and CNS disorders — while simultaneously expanding its lead compound into ophthalmology through fully externally funded studies. This isn't a one-trick biotech. It's a platform play in one of medicine's most underserved mechanistic categories.
The Case for ARTL in One Paragraph
Most coverage of Artelo Biosciences has focused on the glaucoma announcement — a meaningful and capital-efficient pipeline expansion that is genuinely worth tracking. But investors who stop there are missing the larger thesis. Artelo has successfully advanced three separate compounds through human clinical trials, generated positive interim Phase 2 data, attracted academic partners across the UK and Ireland, earned European patent protection extending to 2041, received regulatory guidance from the UK’s MHRA, and drawn unsolicited partnership inquiries from global pharmaceutical companies — all while operating with a lean structure that keeps burn focused on science rather than overhead.
This report goes deeper than the glaucoma headline. We examine all three pipeline assets, the regulatory tailwinds reshaping the cannabinoid therapeutic landscape, the intellectual property architecture that protects the franchise, and the strategic signals embedded in recent SEC filings and press releases.
The Pipeline: Three Compounds, One Scientific Thesis
Artelo’s pipeline is built around a single mechanistic conviction: that modulating lipid-signaling pathways — including the endocannabinoid system — can produce clinically meaningful therapeutic outcomes across multiple disease areas without the CNS liability that has historically limited cannabinoid-based drug development.
That conviction is now backed by human data across all three programs.
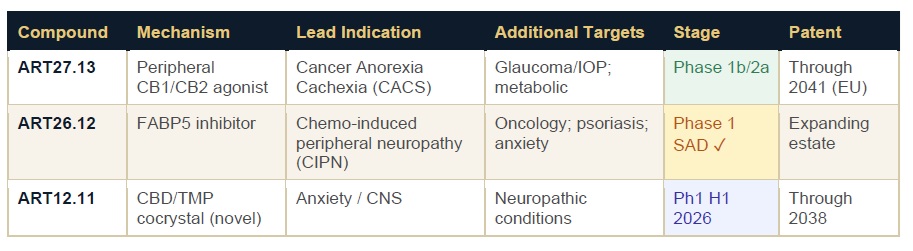
ART27.13 — The Lead Asset with Multiple Shots on Goal
ART27.13 did not originate at Artelo. It was initially developed by AstraZeneca — a meaningful credential that speaks to the compound’s pharmaceutical-grade pedigree. By the time Artelo licensed it, ART27.13 had already been evaluated in seven clinical studies involving more than 280 participants. Artelo’s contribution has been to identify the therapeutic sweet spot: peripheral selectivity that delivers systemic metabolic benefit while sidestepping the CNS effects that made first-generation cannabinoids clinically impractical.
These are not modest improvements. In cancer anorexia cachexia — where there is no FDA-approved treatment — a
The glaucoma expansion layered on top of this is not speculative pivoting. It is a second indication discovered organically through the mechanism: ART27.13’s CB2 receptor activity offers both intraocular pressure modulation and neuroprotective properties that current glaucoma therapies don’t address. The investigator-sponsored trial structure means Artelo advances this indication at zero direct cost to shareholders while retaining full data rights.
“This collaboration broadens ART27.13’s therapeutic profile beyond cancer-related anorexia while preserving our internal focus on the lead indication.” — Gregory D. Gorgas, President & CEO, Artelo Biosciences
ART26.12 — The Hidden Gem in a
If ART27.13 is the headline act, ART26.12 may be the most underappreciated asset in the portfolio. Artelo’s FABP5 (Fatty Acid Binding Protein 5) inhibitor completed its first-in-human Phase 1 single-ascending-dose study in 2025 with results that de-risked the program substantially.
The human data showed no serious adverse events, a favorable safety profile across all dose levels tested up to 1,050 mg, predictable and linear pharmacokinetics, and dosing flexibility in both fed and fasted states. These are the precise attributes a pharmaceutical partner wants to see before committing capital to a Phase 2 collaboration.
What makes ART26.12 strategically significant is what the preclinical data demonstrates beyond the Phase 1 safety signal. In an osteoarthritis pain model, ART26.12 produced sustained analgesic effects without the development of tolerance — a crucial differentiator from opioids and a meaningful advantage over NSAIDs like naproxen, which it matched in efficacy. A separate publication showed antidepressant-like activity on par with sertraline (Zoloft®) with improved cognitive benefits in preclinical models.
The lead indication under an active FDA IND is chemotherapy-induced peripheral neuropathy — a condition affecting a substantial share of the cancer patient population, currently managed with a limited toolkit of off-label medications. Multiple ascending dose enrollment is being prepared for Q3 2026.
FABP inhibitors represent a nascent but rapidly growing pharmacological category. Artelo holds an extensive library of small molecule FABP inhibitors with demonstrated preclinical promise across cancers, neuropathic pain, psoriasis, and anxiety. ART26.12 is the beachhead, not the ceiling, of this franchise.
ART12.11 — A Proprietary CBD Reinvention with Regulatory Momentum
ART12.11 is Artelo’s wholly owned CBD/tetramethylpyrazine cocrystal — a novel crystalline form of cannabidiol that has demonstrated improved pharmacokinetics and efficacy versus conventional CBD in nonclinical studies. The cocrystal format is not a marketing distinction; it is a chemistry distinction that produces measurably different bioavailability and stability profiles.
The compound received favorable UK MHRA regulatory guidance supporting streamlined Phase 1 development with potential accelerated pathways. First-in-human studies are planned for the first half of 2026. Preclinical data showed antidepressant-like activity on par with — and cognitive advantages over — sertraline.
ART12.11 is protected by patents extending to 2038 and is positioned at the intersection of two significant tailwinds: the White House executive order expanding CBD research and the Medicare pilot program for CBD reimbursement announced in late 2025.
The Regulatory Environment Has Shifted in Artelo’s Favor
The cannabinoid therapeutic space has long operated under a regulatory cloud that created genuine commercial uncertainty even for companies with compelling science. That cloud is lifting — and the timing benefits Artelo directly.
- PRESIDENTIAL EXECUTIVE ORDER — CBD RESEARCH EXPANSION
In December 2025, the White House issued an executive order expanding CBD research and access. Artelo specifically highlighted ART12.11 as a leading pharmaceutical-grade cannabidiol candidate positioned to benefit. This policy shift removes a meaningful impediment to clinical development timelines and potential reimbursement conversations with payers.
- MEDICARE CBD REIMBURSEMENT PILOT
The White House launched a Medicare pilot program for CBD reimbursement — a first in U.S. healthcare policy. For a company with a proprietary, pharmaceutical-grade CBD cocrystal (ART12.11) and a peripheral CB1/CB2 agonist in active clinical trials, this policy shift represents a potential inflection point in addressable market size if clinical validation follows the political momentum.
- UK MHRA GUIDANCE — STREAMLINED PATHWAY
Artelo received clear guidance from the UK’s MHRA on a streamlined pathway to the clinic for ART12.11. The UK regulatory environment has historically been more receptive to cannabinoid-adjacent therapeutics, and the MHRA’s accelerated pathway designation positions ART12.11 for faster Phase 1 execution than a U.S.-only development strategy would allow.
These three policy developments — the Presidential EO, the Medicare pilot, and the MHRA pathway — arrived within months of each other. For Artelo, they represent a convergence of regulatory tailwinds that the company was already positioned to capture through a pipeline designed specifically around peripheral selectivity and pharmaceutical-grade formulation.
The Market Opportunity Matrix
Artelo’s total addressable market cannot be evaluated through a single-indication lens. Each of its three programs operates in a distinct therapeutic category with large and growing patient populations.
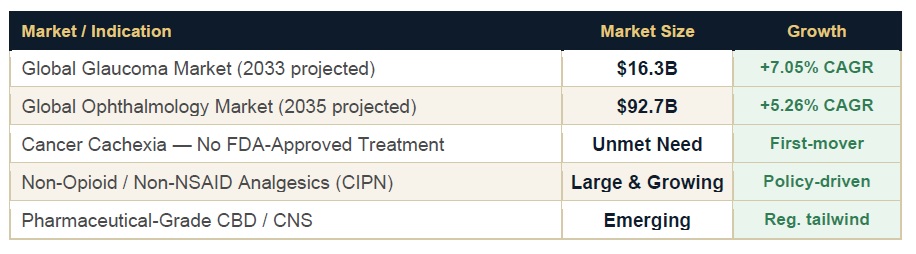
The cancer anorexia cachexia opportunity deserves particular attention because it carries a differentiator that is rarely available in biotech: there is no FDA-approved pharmacological treatment for the condition. The 2020 ASCO guidelines recommend no specific pharmacological intervention as standard of care. ART27.13, if it completes Phase 2 with the signal strength suggested by interim data, would enter a market with no incumbent to displace.
The Intellectual Property Fortress
Patent protection is the commercial runway that converts clinical success into durable value. Artelo’s IP position provides meaningful long-dated protection across all three programs.

The ART27.13 European patent allowance through December 2041 is particularly significant for licensing discussions. A potential pharmaceutical partner evaluating a collaboration would find a compound with 15+ years of commercial patent life, positive Phase 2 human data, and a clean safety profile across 7 studies — the combination that routinely drives outlicensing conversations in mid-stage biotech deals.
The Partnership Signals Investors Should Not Ignore
Among the most consequential disclosures in Artelo’s Q3 2025 earnings update — one that received little investor attention — was the company’s acknowledgment of unsolicited interest:
Following positive Phase 2 results and coverage by UK and USA media outlets, Artelo has received multiple expressions of potential collaboration interest from global and regional pharmaceutical companies. — Artelo Biosciences Q3 2025 Business Update
In the clinical-stage biotech world, unsolicited partnership inquiries at the Phase 2 stage are not routine events. They represent external validation from parties with their own development budgets and scientific advisory teams — parties who don’t make preliminary outreach calls without having reviewed the data themselves.
These expressions of interest have not converted to announced deals, and there is no guarantee they will. But they establish that Artelo’s science is attracting attention beyond the retail investor community — and that the pipeline’s future may not depend solely on Artelo’s own capital position to reach commercialization.
The IST model for the glaucoma study reflects the same dynamic: Professor Augusto Azuara-Blanco at Queen’s University Belfast and the Northern Ireland Clinical Trials Unit did not agree to sponsor a trial of ART27.13 without scientific conviction in the mechanism. Academic institution partnership signals and pharma collaboration inquiries, taken together, suggest that ART27.13’s clinical credibility is resonating well beyond the company’s own press releases.

A Capital-Efficient Strategy Built for the Stage
Artelo’s management team has made a series of deliberate structural decisions that reflect a clear-eyed understanding of how to advance a multi-program biotech pipeline while preserving shareholder capital. The result is a company that is doing more with less — and doing it strategically.
The investigator-sponsored trial model, most visibly deployed for the glaucoma expansion, is the clearest expression of this philosophy. By partnering with Glaucoma UK, the HSC R&D Division, and Queen’s University Belfast, Artelo advances a
The company entered 2026 with a secured equity access facility of up to
The 1-for-3 reverse stock split completed in March 2026 further strengthens the company’s market positioning, improving share price structure and marketability for a broadening investor audience as clinical catalysts approach across all three programs.
As with all clinical-stage biopharmaceutical companies, investment in ARTL involves inherent development, regulatory, and capital risk. Investors are encouraged to review the company’s full SEC filings, including the Annual Report on Form 10-K for fiscal year 2025, for complete financial and risk information. This report does not constitute investment advice. Please see the full disclosure statement below.
What distinguishes Artelo from the broader universe of early-stage biotech is the quality of the science advancing through this stage. Three programs with human data. A lead compound with 280+ clinical participants across seven studies. External institutions willing to co-fund and co-investigate. Global pharmaceutical companies making unsolicited contact. These are the markers of a pipeline that the scientific community is taking seriously — and they represent the foundation on which long-term value creation is built.
Why Artelo Biosciences Warrants Continued Attention
The biotech graveyard is littered with companies that had one interesting compound and ran out of money before the data arrived. Artelo is operating differently — and the structural choices it has made in the past 18 months reflect a management team that understands how to extend runway while advancing science.
The investment thesis rests on several compounding factors:
◆ ART27.13's Phase 2 interim data is positive and statistically meaningful in a disease with no approved standard of care — establishing genuine first-mover potential.
◆ The glaucoma IST advances a
◆ ART26.12 completed a clean, well-tolerated Phase 1 and is progressing to MAD — with preclinical analgesic efficacy on par with naproxen and antidepressant activity matching Zoloft®.
◆ ART12.11, a wholly owned CBD cocrystal with superior PK, is entering Phase 1 with MHRA streamlined guidance and is aligned with the Presidential executive order expanding CBD research.
◆ Unsolicited partnership expressions from global pharmaceutical companies confirm that the science is attracting professional attention beyond the retail market.
◆ European patent protection through 2041 provides a commercial runway long enough to support licensing, partnership, or acquisition discussions from a position of structural strength.
◆ The lean operating structure keeps burn focused on clinical advancement rather than corporate overhead — seven employees,
Artelo is structured for this stage of development — lean, externally leveraged, and advancing on multiple clinical fronts simultaneously. The
But the scientific platform underneath that fragility is not fragile. It is methodical, peer-reviewed, externally validated, and advancing on multiple fronts simultaneously. We will be watching every 2026 milestone closely. The data will tell the real story — and the conditions are in place for that story to be a good one.
IMPORTANT DISCLOSURES
About The Vanderbilt Report: The Vanderbilt Report (AB Holdings LLC) is an independent financial communications platform providing analysis and research on publicly traded companies, with a focus on small-cap and micro-cap equities.
Compensation Disclosure: Artelo Biosciences compensated The Vanderbilt Report in connection with this publication. This report has been prepared for informational purposes only and does not constitute investment advice, a solicitation to buy or sell securities, or a recommendation of any kind. This report contains forward-looking statements based on currently available information; actual results may differ materially.
Risk Warning: Investing in clinical-stage biopharmaceutical companies involves significant risk, including the possible loss of the entire investment. Clinical trials can fail. Regulatory approvals are not guaranteed. Companies discussed herein may face capital constraints, listing compliance issues, or adverse clinical outcomes.
Data sourced from Artelo Biosciences SEC filings (10-K FY2025, 8-K, S-1), GlobeNewswire press releases, artelobio.com pipeline disclosures, and third-party market intelligence reports. All figures accurate as of the date of publication: March 2026.
Media Contact:
The Vanderbilt Report
Jake Rivers
media@vanderbiltreport.com
Photos accompanying this announcement are available at
https://www.globenewswire.com/NewsRoom/AttachmentNg/7dc69ce5-8e3b-4b86-a8d7-367f6cf33918
https://www.globenewswire.com/NewsRoom/AttachmentNg/0bf0b3d0-048d-4b6d-9270-3f83acfd9826
https://www.globenewswire.com/NewsRoom/AttachmentNg/7297ad5f-4c84-4754-ae93-ae532d154f6a
https://www.globenewswire.com/NewsRoom/AttachmentNg/32a1ebb6-12f5-43bc-9a53-584f271d4712

FAQ
What does the fully funded investigator-sponsored glaucoma study mean for ARTL shareholders?
How meaningful is ART27.13’s Phase 2 interim result for ARTL (NASDAQ: ARTL)?
What is the development status of ART26.12 and its near-term timeline for ARTL?
How does ART12.11’s MHRA guidance and U.S. policy affect ARTL’s CBD program?
What are the near-term capital considerations for ARTL investors following the March 2026 update?







