Immuron Double Digit 3Q Sales Growth
Rhea-AI Summary
Immuron (NASDAQ: IMRN) reported unaudited Q3 sales of AUD$1.5 million, up 16% on the prior comparative period, with YTD Mar 2026 sales of AUD$5.7 million up 7%.
By region: Australia Q3 AUD$0.9M (+15%; YTD AUD$4.2M +14%), USA Q3 AUD$0.5M (+1%; YTD AUD$1.3M +10%), Canada Q3 AUD$0.1M (Q3 +82% vs prior quarter; YTD AUD$0.1M -65%). Growth attributed to expanded digital marketing, promotions, new store listings and increased travel.
Positive
- Global Q3 sales +16% to AUD$1.5 million
- Australia YTD sales +14% to AUD$4.2 million
- U.S. YTD sales +10% to AUD$1.3 million
- Canada Q3 sales +82% versus prior quarter
Negative
- Canada YTD sales down 65% to AUD$0.1 million
- Australian dollar strength weighed on U.S. growth
News Market Reaction – IMRN
On the day this news was published, IMRN gained 13.35%, reflecting a significant positive market reaction. Argus tracked a peak move of +13.8% during that session. Our momentum scanner triggered 10 alerts that day, indicating notable trading interest and price volatility. This price movement added approximately $962K to the company's valuation, bringing the market cap to $8.17M at that time. Trading volume was exceptionally heavy at 8.8x the daily average, suggesting very strong buying interest.
Data tracked by StockTitan Argus on the day of publication.
Key Figures
Market Reality Check
Peers on Argus
IMRN was up 2.42% while momentum-screened biotech peers like PHGE and BRTX were down (sector median about -4.9%). This points to a stock-specific reaction to the Travelan sales update rather than a broad sector move.
Historical Context
| Date | Event | Sentiment | Move | Catalyst |
|---|---|---|---|---|
| Mar 05 | HY26 results update | Positive | +4.5% | First-half FY26 results, revenue growth, cash runway into 2027 and pipeline progress. |
| Mar 05 | Conference presentation | Neutral | +4.5% | CEO scheduled virtual presentation at Coffee Microcaps Conference with investor materials. |
| Feb 25 | HY26 results reset | Positive | +17.1% | Half-year 2026 sales growth, narrowed loss, cash position and strategic reset toward partnerships. |
| Jan 22 | Research flash update | Positive | +1.4% | Analyst flash update on H1 FY26 revenue with maintained Buy-Extended rating and target. |
| Jan 19 | Sales growth update | Positive | -7.8% | H1 FY26 Travelan sales growth across regions with ProIBS launch in Australia. |
Recent fundamental and research updates have generally seen positive price alignment, with only one notable selloff on a prior sales-growth release.
Over the last few months, Immuron has repeatedly highlighted Travelan-driven growth and gradual financial improvement. HY26 global sales reached AUD$4.2 million, up 5% year-on-year, with Australian and U.S. sales both growing double digits and cash rising to about AUD$10.0 million. Research notes maintained bullish ratings and price targets while emphasizing revenue traction and the clinical pipeline. A January sales update showing continued growth still saw a -7.83% move, but later HY26 and strategy news on Feb 25 produced a 17.14% gain, suggesting the market has lately rewarded execution updates similar to this Q3 sales release.
Market Pulse Summary
The stock surged +13.3% in the session following this news. A strong positive reaction aligns with ongoing evidence of steady Travelan sales expansion. Prior HY26 and research updates with revenue growth triggered gains of up to 17.14%, while one earlier sales release saw a -7.83% pullback. Investors have previously weighed execution progress against funding history and dilution from capital raises. Any sharp upside following this Q3 data could be vulnerable if growth moderates in later quarters or if additional equity financing re-enters focus.
Key Terms
gastrointestinal (GI) tract medical
AI-generated analysis. Not financial advice.
Sales Highlights (unaudited):
| Global |
|
| Australia |
|
| Canada |
|
| USA |
|
MELBOURNE, Australia, April 17, 2026 (GLOBE NEWSWIRE) -- Immuron Limited (ASX: IMC; NASDAQ: IMRN), an Australian based and globally integrated biopharmaceutical company is pleased to announce continued sales growth (unaudited) of Travelan®, an over-the-counter immune supplement that targets pathogenic bacteria and the toxins they produce in the gastrointestinal (GI) tract.
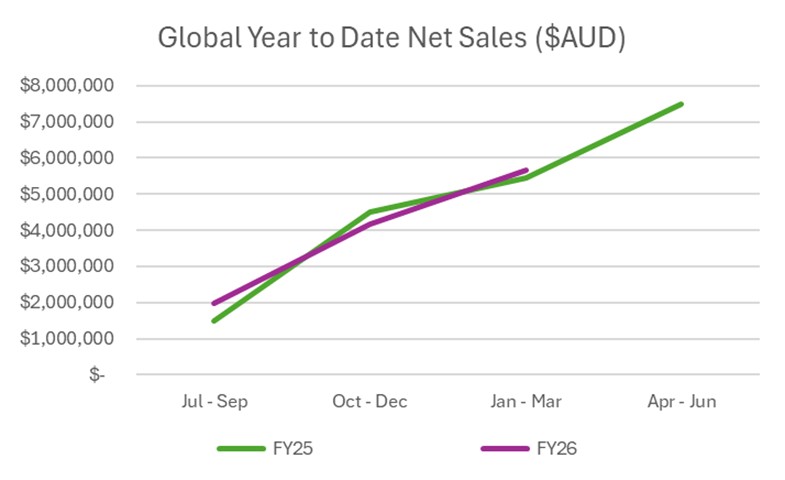
Continued Travelan® Q3 sales growth (+
Q3 sales in the U.S. increased (+
During FY25 we had a Q1 pipeline fill into over a thousand Canada retail doors on the back of securing listings within key pharmacy and grocery retail groups. We previously reported that sales picked up on the back of consumer promotions in Q2 FY26 (+
This release has been authorised by the directors of Immuron Limited.
COMPANY CONTACT:
Steven Lydeamore
Chief Executive Officer
steve@immuron.com
About Travelan®
Travelan® is an orally administered passive immunotherapy that prophylactically reduces the likelihood of contracting travelers’ diarrhea, a digestive tract disorder that is commonly caused by pathogenic bacteria and the toxins they produce. Travelan® is a purified tablet preparation of hyper-immune bovine antibodies and other factors, which when taken with meals bind to diarrhea-causing bacteria and prevent colonization and the pathology associated with traveler’s diarrhea. In Australia, Travelan® is a listed medicine on the Australian Register for Therapeutic Goods (AUST L 106709) and is indicated to reduce the risk of Traveler’s Diarrhea, reduce the risk of minor gastro-intestinal disorders and is antimicrobial. In Canada, Travelan® is a licensed natural health product (NPN 80046016) and is indicated to reduce the risk of Traveler’s Diarrhea. In the U.S., Travelan® is sold as a dietary supplement for digestive tract protection.
About Immuron
Immuron Limited (ASX: IMC, NASDAQ: IMRN), is an Australian biopharmaceutical company focused on developing and commercializing orally delivered targeted polyclonal antibodies for the treatment of inflammatory mediated and infectious diseases.
Immuron Platform Technology
Immuron’s proprietary technology is based on polyclonal immunoglobulins (IgG) derived from engineered hyper-immune bovine colostrum. Immuron has the capability of producing highly specific immunoglobulins to any enteric pathogen and our products are orally active. Bovine IgG can withstand the acidic environment of the stomach and is resistant to proteolysis by the digestive enzymes found in the Gastrointestinal (GI) tract. Bovine IgG also possesses this unique ability to remain active in the human GI tract delivering its full benefits directly to the bacteria found there. The underlying nature of Immuron’s platform technology enables the development of medicines across a large range of infectious diseases. The platform can be used to block viruses or bacteria at mucosal surfaces such as the Gastrointestinal tract and neutralize the toxins they produce.
For more information visit: https://www.immuron.com.au/ and https://www.travelan.com
Sign up to Immuron’s Investor Hub: Here
FORWARD-LOOKING STATEMENTS:
This press release may contain “forward-looking statements” within the meaning of Section 27A of the Securities Act of 1933 and Section 21E of the Securities Exchange Act of 1934, each as amended. Such statements include, but are not limited to, any statements relating to our growth strategy and product development programs and any other statements that are not historical facts. Forward-looking statements are based on management’s current expectations and are subject to risks and uncertainties that could negatively affect our business, operating results, financial condition and stock value. Factors that could cause actual results to differ materially from those currently anticipated include: risks relating to our growth strategy; our ability to obtain, perform under and maintain financing and strategic agreements and relationships; risks relating to the results of research and development activities; risks relating to the timing of starting and completing clinical trials; uncertainties relating to preclinical and clinical testing; our dependence on third-party suppliers; our ability to attract, integrate and retain key personnel; the early stage of products under development; our need for substantial additional funds; government regulation; patent and intellectual property matters; competition; as well as other risks described in our SEC filings. We expressly disclaim any obligation or undertaking to release publicly any updates or revisions to any forward-looking statements contained herein to reflect any change in our expectations or any changes in events, conditions or circumstances on which any such statement is based, except as required by law.
A photo accompanying this announcement is available at https://www.globenewswire.com/NewsRoom/AttachmentNg/91da4c25-bccc-44c5-bc67-81258bbb1ba5

