Kane Biotech Presents revyve® Clinical Data at Symposium on Advanced Wound Care (SAWC) Spring Conference
Rhea-AI Summary
Kane Biotech (OTC:KNBIF) presented case-study clinical data for revyve at the SAWC Spring conference (April 8-12, 2026).
Highlights include >20-year venous leg ulcers reduced 85% at 33 weeks, complete pain elimination by week 6, 90% wound volume reduction in seven months for a tunneling wound, and wound pH falling from 8.0 to 6.8 in four weeks.
Positive
- 85% wound size reduction at 33 weeks in a >20-year venous leg ulcer case
- Complete pain elimination by week 6 for the long-standing VLU patient
- 90% wound volume reduction in seven months for a deep tunneling wound
- pH normalization from 8.0 to 6.8 in four weeks with associated size reductions
Negative
- Findings presented are case studies and small patient samples, limiting broad generalizability
- No randomized controlled trial outcomes or large cohort data were reported at SAWC
- >20 year‑old venous leg ulcers show dramatic healing and pain elimination with revyve
- Thermoreversible revyve transforms deep tunneling wound care, delivering
90% wound volume reduction - revyve rapidly normalizes chronic wound pH, driving accelerated healing in weeks
WINNIPEG, Manitoba, April 16, 2026 (GLOBE NEWSWIRE) -- Kane Biotech Inc. (TSX-V:KNE) (“Kane Biotech”, “Kane” or the “Company”) today announces that Dr. Miloslav Sailer, External R&D Leader, presented at the Symposium on Advanced Wound Care (“SAWC”) Spring conference held from April 8-12, 2026 in Charlotte, North Carolina.
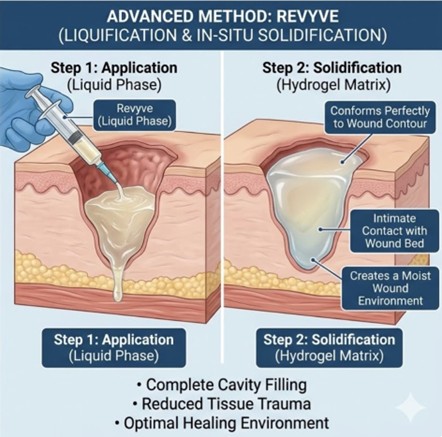
At this year’s conference, Kane Biotech shared case study data on how revyve can help patients with complex chronic non-healing as well as clinical mechanistic investigations. The link to the presentations can be found here and include:
- A Patient with >20-year-old Venous Leg Ulcers (VLUs) with venous insufficiency, chronic phlebolymphedema, extreme pain, and a combined wound size of >120 cm2 was converted to revyve treatment. After 11 weeks the wound size was reduced by
40% with complete elimination of pain by week 6. Follow-up at 33 weeks demonstrated85% wound size reduction. - A quadriplegic patient with a 1.5-year-old deep tunneling wound switched from packing the wound with impregnated gauze, to filling the contours of the cavity using the thermoreversible technology of revyve. The patient converted to a healing trajectory with significant pain reduction and
90% wound volume reduction in 7 months. - Mechanistic investigations of revyve showed the chronic wound average pH dropped from a pH 8.0 to a pH 6.8 in 4 weeks with an associated
45% average wound size reduction in 4 weeks for two patients and a60% wound size reduction in 1 week with a third patient.
SAWC is a multidisciplinary community of wound care professionals — including physicians, nurses, podiatrists, physical therapists, researchers, administrators and wound care companies — that share the latest evidence-based practices, clinical innovations, and research aimed at improving outcomes in wound management.
About Kane Biotech Inc. (TSX-V:KNE)
Kane Biotech is commercializing and developing novel wound care treatments that disrupt biofilms and transform healing outcomes. Biofilms are one of the main contributors to antibiotic resistance in wounds, resulting in serious clinical outcomes and significant cost. revyve addresses both biofilms and wound bacteria. revyve Antimicrobial Wound Gel, revyve Antimicrobial Wound Gel Spray and revyve Antimicrobial Skin and Wound Cleanser are all U.S. FDA 510(k) cleared. revyve Antimicrobial Wound Gel and revyve Antimicrobial Wound Gel Spray are also Health Canada approved. To learn more, visit revyvegel.com or revyvegel.ca.
Join Kane’s Distribution List & Social Media:
To stay informed on the latest developments, sign up for the Company’s email distribution list here.
Follow Kane
Website: kanebiotech.com
LinkedIn: https://www.linkedin.com/company/kanebiotech/
Presentation: Disrupting Biofilms to Save Limbs and Transform Wound Care
For more information:
| Dr. Robert Huizinga | Ray Dupuis |
| Interim CEO | Chief Financial Officer |
| Kane Biotech Inc. | Kane Biotech Inc. |
| rhuizinga@kanebiotech.com | rdupuis@kanebiotech.com |
| (780) 970-1100 | (204) 298-2200 |
Neither the TSX Venture Exchange nor its Regulation Services Provider (as that term is defined in policies of the TSX Venture Exchange) accepts responsibility for the adequacy or accuracy of this release.
Caution Regarding Forward-Looking Information
This press release contains certain statements regarding Kane Biotech Inc. that constitute forward-looking information under applicable securities law. These statements reflect management’s current beliefs and are based on information currently available to management. Certain material factors or assumptions are applied in making forward-looking statements, and actual results may differ materially from those expressed or implied in such statements. These risks and uncertainties include, but are not limited to, risks relating to the Company’s: (a) financial condition, including lack of significant revenues to date and reliance on equity and other financing; (b) business, including its early stage of development, government regulation, market acceptance for its products, rapid technological change and dependence on key personnel; (c) intellectual property including the ability of the Company to protect its intellectual property and dependence on its strategic partners; and (d) capital structure, including its lack of dividends on its common shares, volatility of the market price of its common shares and public company costs. Further information about these and other risks and uncertainties can be found in the disclosure documents filed by the Company with applicable securities regulatory authorities, available at www.sedarplus.ca. The Company cautions that the foregoing list of factors that may affect future results is not exhaustive.
An image accompanying this announcement is available at https://www.globenewswire.com/NewsRoom/AttachmentNg/a7606fbc-273a-4100-a03b-cc73a0d8cd61

