Lexaria's Oral GLP-1 Drug Strategy Validated by Industry
Rhea-AI Summary
Lexaria (NASDAQ:LEXX) outlines its strategy to develop oral GLP-1 capsule and tablet alternatives using its patented DehydraTECH platform, citing intellectual property established in 2024–2025 and early-stage commercial discussions with several multinational pharmaceutical companies. The company highlights market forecasts suggesting oral GLP-1s could represent a $40–45 billion annual market within four years and cites patient tolerability and discontinuation rates as drivers for its program.
Positive
- Established DehydraTECH IP in 2024 and 2025
- Early-stage discussions with several multinational pharmaceutical companies
- Market forecasts imply a $40–45B annual oral GLP-1 opportunity within four years
- Oral GLP-1 share expected to rise from 5% (2025) to 33% by 2030
Negative
- High GI side-effect rates: 74% (injected semaglutide) and 80% (oral semaglutide) in cited studies
- High discontinuation: 46.5% of patients with and 64.8% without type 2 diabetes stopped GLP-1 use within 1 year
- Pfizer discontinued danuglipron Phase III after a safety signal (elevated liver enzymes), illustrating safety risks for oral candidates
Market Reaction – LEXX
Following this news, LEXX has gained 9.45%, reflecting a notable positive market reaction. Our momentum scanner has triggered 6 alerts so far, indicating moderate trading interest and price volatility. The stock is currently trading at $0.80. This price movement has added approximately $2M to the company's valuation.
Data tracked by StockTitan Argus (15 min delayed). Upgrade to Silver for real-time data.
Key Figures
Market Reality Check
Peers on Argus
LEXX is up 2.34% while key biotech peers are mixed: ASBP up 22.28%, CING down 3.08%, CRIS down 7.53%, PMN down 0.22%, and NRXS down 6.04%. Scanner momentum shows only a couple of names moving up, pointing to a stock-specific reaction rather than a broad sector rotation.
Historical Context
| Date | Event | Sentiment | Move | Catalyst |
|---|---|---|---|---|
| Mar 04 | R&D program update | Positive | +5.0% | Announced 2026 human and animal GLP-1 studies to expand data and IP. |
| Feb 05 | Clinical study results | Positive | -8.7% | Human Pilot Study #5 showed fewer adverse events for oral DHT-liraglutide. |
| Jan 22 | Patent portfolio expansion | Positive | +2.1% | Six new patents lifted total to 60 across major global markets. |
| Jan 12 | Annual CEO letter | Positive | +3.2% | CEO outlined Phase 1b success, AE reductions and financing progress. |
| Dec 30 | Phase 1b data update | Positive | -2.9% | Additional GLP-1-H24-4 results confirmed safety and AE reductions vs Rybelsus. |
Recent GLP-1 and IP updates have produced mixed reactions, with both gains and pullbacks following clinically and strategically positive news.
Over the past few months, Lexaria has steadily advanced its DehydraTECH GLP‑1 strategy. Updates included new 2026 R&D plans with a 5‑week human study and two large animal studies (Mar 4, 2026), positive final results from Human Pilot Study #5 showing reduced adverse events (Feb 5, 2026), and six additional patents bringing the total to 60 (Jan 22, 2026). Earlier, an annual CEO letter highlighted Phase 1b success with a 48% adverse‑event reduction versus Rybelsus® and ongoing financing activity. Today’s industry-validation update fits this trajectory of building clinical evidence and IP to support partnering.
Market Pulse Summary
The stock is up +9.4% following this news. A strong positive reaction aligns with Lexaria’s positioning in a rapidly expanding oral GLP‑1 market projected at $40–$45 billion annually within a few years. Recent history shows both rallies and pullbacks after positive GLP‑1 data, so durability often depended on follow‑through milestones such as clinical readouts, patents, and partnering updates rather than narrative alone. Investors have also reacted in the past to financing needs and dilution, which remained ongoing considerations in regulatory filings.
Key Terms
glp-1 medical
gastrointestinal medical
phase i medical
phase ii medical
phase iii medical
adverse events medical
fda regulatory
AI-generated analysis. Not financial advice.
"The window to shape the oral GLP-1 market is open now...."
Early-stage discussions with several multinational pharmaceutical companies currently underway
KELOWNA, BC / ACCESS Newswire / March 24, 2026 / Lexaria Bioscience Corp. (NASDAQ:LEXX), (the "Company" or "Lexaria"), a global innovator in drug delivery platforms provides this update of developments within the glucagon-like peptide-1 ("GLP-1") market.
Lexaria has been listening and responding to the concerns of those using GLP-1 drugs, who overwhelmingly indicate their preference for oral versus injected delivery and who are desperately seeking safer options whose side effects do not make them feel sick. This has driven Lexaria's mission to develop better tolerated oral GLP-1 capsule and tablet alternatives. After far-reaching proprietary formulation advancements and the establishment of intellectual property in 2024 and 2025, Lexaria is currently developing additional enhancements to its DehydraTECH GLP-1 platform that we are hopeful could instigate commercial development with leading industry partners.
Lexaria first began reporting on the momentum within the fast-moving GLP-1 drug sector during 2023. Growth in the sector has been, if anything, even more significant than we anticipated at the time and has supported our business strategy of pursuing opportunities within the GLP-1 market. Examples of this growth and the challenges abound:
Eli Lilly's tirzepatide is expected to be the #1-selling drug in the world for 2026, with revenue estimated at
Injected GLP-1 drugs often produce very similar results to oral GLP-1 drugs. For instance, in a 2021 study, patients who took once-weekly semaglutide injections lost an average of ~
In a very large 6-year study of over 125,000 patients, it was reported that
From a strategic perspective, Lexaria understood that losing roughly half your customers could not have been a welcome development for the manufacturers of GLP-1 drugs. That's why we focused on the goals of improving patient experiences via the preferred oral delivery method whilst preserving the efficacy of the underlying medication. To do so, we had to better understand the needs of the market to bolster our business case.
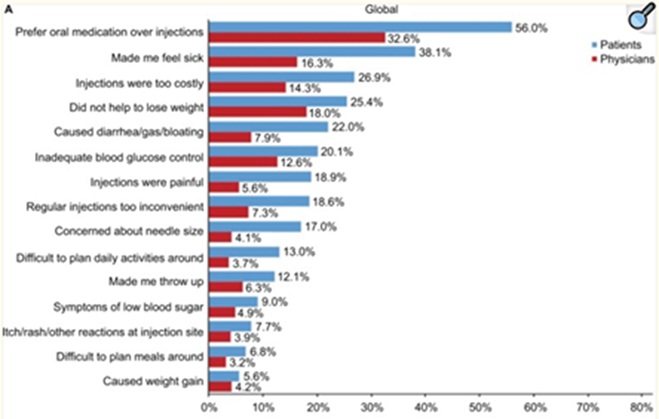
In a survey of 2,173 patients who discontinued the use of GLP-1 drugs, a majority of the patients identified two main areas of concern: the GI-related discomfort or the negativity associated with injections:
In a separate survey published in March 2026, of 1,536 Canadians and 1,012 Americans, Leger Healthcare market research reported a number of key findings related to GLP-1 use and intentions across North America:
11% of American adults and8% of Canadian adults are currently taking a GLP-1 prescriptionExtrapolated, this suggests that 28M+ American adults and 3M+ Canadian adults are currently using GLP-1 prescriptions
12% of Americans and6% of Canadians are interested in taking a GLP-1 prescription71% of Americans and58% of Canadians cite weight loss as a primary reason for their interest in GLP-1 drugs
Given that nearly two-thirds of GLP-1 customers focused on weight loss tend to discontinue use of the drugs within the first year, Lexaria knew that our attempts to improve the patient experience could provide a profoundly positive business case. With less than
By the beginning of 2025, the sector was buzzing with anticipation of Pfizer's® orally-delivered GLP-1 candidate, danuglipron, which had shown great promise in Phase I and II studies. Then on April 14, 2025, Pfizer reported that they were discontinuing development of danuglipron because in an ongoing Phase III study of 1,400 people, one person experienced elevated liver enzymes that resolved after its discontinuation. Pfizer had little choice but to forego a potential multi-billion-dollar drug blockbuster because the adverse events were simply not acceptable. This was a devastating blow to Pfizer and to all those hoping for a new oral alternative in GLP-1.
But industry development of GLP-1 drugs has not ended. If anything, it has only intensified. In May of 2025, it was reported that 39 new GLP-1 medications were under development. Companies such as Viking Therapeutics® and Structure Therapeutics® are both pursuing oral GLP-1 drug candidates.
Similarly, Eli Lilly is developing orforglipron, a small-molecule GLP-1 candidate. The New England Journal of Medicine® published an article detailing the outcomes of orforglipron, being investigated as an oral treatment for obesity. This Phase III multinational study provided average weight loss of up to
While orforglipron has shown in studies to be slightly more effective at reducing body weight and reducing blood sugar levels than semaglutide, "Orforglipron showed higher rates of adverse side effects such as nausea, vomiting and other gastrointestinal issues compared with semaglutide. More people discontinued orforglipron during the trial than those who stopped taking semaglutide, too." (Scientific American®)
Novo Nordisk's semaglutide received FDA approval for an oral tablet for weight loss in January 2026 and reached over 26,000 prescriptions within two weeks of launch. Novo Nordisk expects oral GLP-1 tablets to represent over
These forecasts imply that oral GLP-1 markets for all conditions could represent a
According to PRC,® "the window to shape the oral GLP‑1 market is open now-and it will not remain open for long. Decisions made today across manufacturing and commercialisation will determine which companies are best positioned as these therapies move from promise to scale. As the landscape evolves rapidly, early, deliberate investment in the right capabilities and strategic partnerships will be critical to securing a leadership position."
Lexaria is executing its strategy of developing must-have technology that delivers solutions to meet patient's #1 requests: an effective GLP-1 drug delivered orally with reduced side effects.
Our 2026 R&D and business development plans are designed to foster commercial relationships with pharmaceutical companies around the world, including those located in heavily populated countries such as India, China, and Brazil where key semaglutide patents expire in 2026, opening new opportunities for generic GLP-1 candidates utilizing Lexaria's patented DehydraTECH technology. Lexaria is currently in early-stage discussions with several multinational pharmaceutical companies related primarily but not exclusively to our work in GLP-1.
About Lexaria Bioscience Corp. & DehydraTECH
DehydraTECH™ is Lexaria's patented drug delivery formulation and processing platform technology which improves the way a wide variety of drugs enter the bloodstream, always through oral delivery. DehydraTECH has repeatedly evidenced the ability to increase bio-absorption, reduce side-effects, and deliver some drugs more effectively across the blood brain barrier. Lexaria operates a licensed in-house research laboratory and holds a robust intellectual property portfolio with over 60 patents granted and additional patents pending worldwide. For more information, please visit www.lexariabioscience.com.
CAUTION REGARDING FORWARD-LOOKING STATEMENTS
This press release includes forward-looking statements. Statements as such term is defined under applicable securities laws. These statements may be identified by words such as "anticipate," "if," "believe," "plan," "estimate," "expect," "intend," "may," "could," "should," "will," and other similar expressions. Such forward-looking statements in this press release include, but are not limited to, statements by the Company relating to the intended use of proceeds from the offering and relating to the Company's ability to carry out research initiatives, receive regulatory approvals or grants or experience positive effects or results from any research or study. Such forward-looking statements are estimates reflecting the Company's best judgment based upon current information and involve a number of risks and uncertainties, and there can be no assurance that the Company will actually achieve the plans, intentions, or expectations disclosed in these forward-looking statements. As such, you should not place undue reliance on these forward-looking statements. Factors which could cause actual results to differ materially from those estimated by the Company include, but are not limited to, market and other conditions, government regulation and regulatory approvals, managing and maintaining growth, the effect of adverse publicity, litigation, competition, scientific discovery, the patent application and approval process, potential adverse effects arising from the testing or use of products utilizing the DehydraTECH technology, the Company's ability to maintain existing collaborations and realize the benefits thereof, delays or cancellations of planned R&D that could occur related to pandemics or for other reasons, and other factors which may be identified from time to time in the Company's public announcements and periodic filings with the US Securities and Exchange Commission on EDGAR. The Company provides links to third-party websites only as a courtesy to readers and disclaims any responsibility for the thoroughness, accuracy or timeliness of information at third-party websites. There is no assurance that any of Lexaria's postulated uses, benefits, or advantages for the patented and patent-pending technology will in fact be realized in any manner or in any part. No statement herein has been evaluated by the Food and Drug Administration (FDA). Lexaria-associated products are not intended to diagnose, treat, cure or prevent any disease. Any forward-looking statements contained in this release speak only as of the date hereof, and the Company expressly disclaims any obligation to update any forward-looking statements or links to third-party websites contained herein, whether as a result of any new information, future events, changed circumstances or otherwise, except as otherwise required by law.
INVESTOR CONTACT:
George Jurcic - Head of Investor Relations
ir@lexariabioscience.com
Phone: 250-765-6424, ext 202
SOURCE: Lexaria Bioscience Corp.
View the original press release on ACCESS Newswire
FAQ
What is Lexaria announcing about its oral GLP-1 plans (LEXX) on March 24, 2026?
How large could the oral GLP-1 market be for Lexaria (LEXX) by 2030–2031?
Why does Lexaria (LEXX) emphasize oral delivery and tolerability for GLP-1 drugs?
What safety setback in oral GLP-1 development does Lexaria reference in its March 24, 2026 update?
Does Lexaria (LEXX) have patents or IP supporting its oral GLP-1 approach?
How do patient discontinuation rates affect Lexaria's business case for oral GLP-1s (LEXX)?







