Adia Nutrition Announces Submission of Final Revised Protocol for Lower Back Pain Study with AdiaVita
Rhea-AI Summary
Adia Nutrition (OTCQB: ADIA) announced that subsidiary Adia Med submitted a final revised protocol to the Institutional Review Board on March 3, 2026, for a lower back pain study of AdiaVita.
The trial is a single-blind, placebo-controlled IV study randomizing ~100 participants with a three-month initial treatment and a crossover phase; patient-pay remains $5,000 per participant.
AI-generated analysis. Not financial advice.
Positive
- Final revised protocol submitted to IRB (March 3, 2026)
- Trial design: ~100 participants, single-blind, placebo-controlled
- Crossover phase provides within-subject additional data
- Non-invasive systemic IV delivery as alternative to injections
Negative
- Study approval still pending IRB decision
- Patient-pay model charges $5,000 per participant
News Market Reaction – ADIA
On the day this news was published, ADIA declined 4.12%, reflecting a moderate negative market reaction.
Data tracked by StockTitan Argus on the day of publication.
Winter Park, Florida--(Newsfile Corp. - March 3, 2026) - Adia Nutrition Inc. (OTCQB: ADIA), a company focused on regenerative medicine and therapeutic solutions, today announced that its subsidiary, Adia Med of Winter Park, LLC, has submitted its final revised protocol to the Institutional Review Board (IRB) for the lower back pain clinical study.
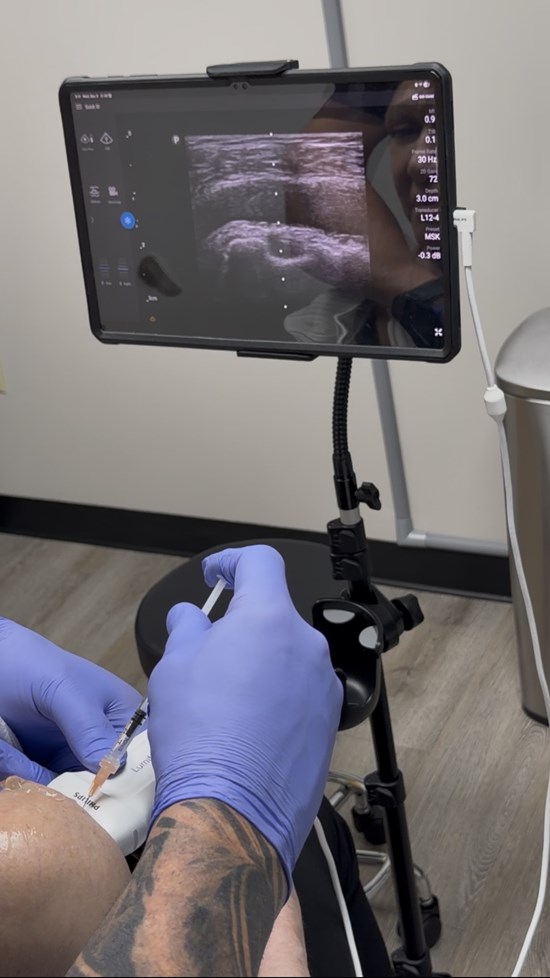
To view an enhanced version of this graphic, please visit:
https://images.newsfilecorp.com/files/10520/286105_img_8679_1.jpg
Following the initial protocol submission on February 16, 2026, the IRB provided constructive feedback with positive comments on the overall study design and approach. The requested changes were minor in nature, primarily relating to refinements in study design details, patient safety measures, and outcome assessment parameters. Adia Med has incorporated these suggestions and submitted the final draft, which the Company believes addresses the IRB's input effectively and positions the protocol for approval.
The key features of the study remain consistent:
- A single-blind, placebo-controlled trial randomizing approximately 100 participants into two groups: one receiving AdiaVita via intravenous (IV) administration and the other receiving a placebo.
- Primary objective: To evaluate AdiaVita's potential to reduce inflammation and support repair of damaged tissue in the lower back.
- Crossover design: Following the initial three-month treatment period, participants in the placebo group will receive AdiaVita in the subsequent phase for additional within-subject data and extended follow-up.
- Non-invasive systemic delivery via IV infusion, intended as a less invasive alternative to conventional targeted injections.
The patient-pay model remains
This development continues to build on the progress of Adia Med's IRB-approved 24-month Autism Spectrum Disorder (ASD) study (ClinicalTrials.gov NCT07304440), which is actively recruiting children aged 3-12 from the United States and internationally. That trial examines the combination of AdiaVita with glutathione therapy compared to glutathione alone, with participants receiving AdiaVita treatments three times during the interventional phase.
"We appreciate the IRB's positive and detailed feedback on the initial draft, which has helped us refine and strengthen the protocol," said Larry Powalisz, CEO of Adia Nutrition. "With the final revised version now submitted, we are optimistic about receiving approval soon and moving forward with this important study on AdiaVita's potential in managing lower back pain through straightforward IV delivery. Recruitment for the lower back pain study will be announced shortly after final IRB approval and registration on ClinicalTrials.gov."
For information on the ongoing ASD study or to apply, please visit adiamed.com or ClinicalTrials.gov (NCT07304440).
For questions, inquiries, or additional information, please contact Larry Powalisz at ceo@adiamed.com or by phone at 321-788-0850.
Clinic owners and healthcare practitioners interested in licensing the Adia Med name or integrating Adia's regenerative therapies into their practice are encouraged to reach out directly. Strategic partnerships are welcomed as part of Adia's continued mission to expand access to advanced stem cell solutions.
About ADIA Nutrition Inc.:
Adia Nutrition Inc. (OTCQB: ADIA), based in Winter Park, Florida, is a publicly traded company advancing healthcare through innovation. The company specializes in sales of stem cell and regenerative products, such as AdiaVita and AdiaLink, through its lab division, Adia Labs LLC, which is expanding to include insurance-billable wound care products. Adia is also growing nationwide with Adia Med clinics, specializing in orthopedic, pain management, and wound repair. Adia Med clinics also offer specialized regenerative treatments like stem cell therapies and platelet-rich plasma (PRP), advanced treatments including therapeutic plasma exchange (TPE) and autologous hematopoietic stem cell transplantation (aHSCT), and wound repair services.
Revenue is generated through service fees, product sales, equity stakes, and billing insurance for healthcare treatments. Additionally, Adia Nutrition Inc. invests in aligned businesses such as Cement Factory LLC, a nutrition and supplement company with shared values and a focus on health and wellness. Through bold partnerships with top-tier medical entities and unwavering dedication to standardized, FDA-approved lab protocols, Adia Nutrition Inc. is revolutionizing healthcare, igniting a nationwide movement to empower communities with groundbreaking regenerative solutions and vibrant, holistic wellness.
Website: www.adianutrition.com
Website: www.adiamed.com
Website: www.adialabs.com
Website: www.cementfactory.co
Twitter (X): @ADIA_Nutrition
Safe Harbor: This Press Release contains forward-looking statements within the meaning of Section 27A of the Securities Act of 1933 and Section 21E of the Securities Exchange Act of 1934. These forward-looking statements are based on the current plans and expectations of management and are subject to a few uncertainties and risks that could significantly affect the company's current plans and expectations, as well as future results of operations and financial condition. A more extensive listing of risks and factors that may affect the company's business prospects and cause actual results to differ materially from those described in the forward-looking statements can be found in the reports and other documents filed by the company with the Securities and Exchange Commission and OTC Markets, Inc. OTC Disclosure and News Service. The company undertakes no obligation to publicly update or revise any forward-looking statements, because of new information, future events or otherwise.

To view the source version of this press release, please visit https://www.newsfilecorp.com/release/286105
