Actinium Pharmaceuticals, Inc. Announces Compelling Pan-Tumor Data for ATNM-400 Demonstrating Broad Efficacy Across Prostate, Lung, and Breast Cancer Models at the 2026 American Association of Cancer Research Annual Meeting
Rhea-AI Summary
Actinium Pharmaceuticals (NYSE: ATNM) presented preclinical AACR 2026 data showing ATNM-400, an Ac-225 radioconjugate, with pan-tumor activity in prostate, EGFR-mutant lung, and HER2+ breast cancer models.
Data show efficacy in low/negative PSMA prostate models, superior activity versus several approved ADCs and targeted agents, durable control post-treatment in breast models, and no in vivo toxicities at therapeutic doses.
Positive
- None.
Negative
- None.
News Market Reaction – ATNM
On the day this news was published, ATNM declined 10.86%, reflecting a significant negative market reaction. Argus tracked a peak move of +4.4% during that session. Argus tracked a trough of -22.6% from its starting point during tracking. Our momentum scanner triggered 12 alerts that day, indicating notable trading interest and price volatility. This price movement removed approximately $6M from the company's valuation, bringing the market cap to $47.69M at that time. Trading volume was elevated at 2.8x the daily average, suggesting increased selling activity.
Data tracked by StockTitan Argus on the day of publication.
Key Figures
Market Reality Check
Peers on Argus
ATNM gained 5.56% while peers showed mixed moves (e.g., PDSB +2.29%, CUE -20.56%, GNTA -4.87%). No peers appeared in momentum scanners, indicating a stock-specific reaction to the ATNM-400 AACR data rather than a broad biotech move.
Historical Context
| Date | Event | Sentiment | Move | Catalyst |
|---|---|---|---|---|
| Apr 06 | AACR data preview | Positive | +11.2% | Announced two AACR 2026 abstracts with new ATNM-400 and Actimab-A data. |
| Feb 17 | Conference notice | Positive | -2.7% | Flagged upcoming AACR presentations for ATNM-400 and Actimab-A without new data. |
| Dec 12 | Breast cancer data | Positive | +4.1% | Presented SABCS 2025 data showing potent ATNM-400 activity in breast subtypes. |
| Dec 01 | TNBC preclinical data | Positive | -0.7% | Announced TNBC and resistance-overcoming efficacy data for ATNM-400 at SABCS. |
| Nov 17 | Strategy feature | Positive | +14.2% | Article on ATNM-400 prioritization and competitive position in radiotherapy market. |
ATNM-400 news has often led to positive moves, but there are notable instances of negative or muted reactions, indicating inconsistent trading responses to similar preclinical updates.
Over the past six months, Actinium has repeatedly highlighted preclinical progress for ATNM-400 across solid tumors and broader radiotherapy strategy. AACR- and SABCS-linked releases on ATNM-400 pan-tumor activity and breast cancer data produced mixed but often positive moves, including reactions of +11.18% and +14.17%. Other conference and abstract notices saw smaller or negative reactions. Today’s detailed AACR 2026 pan-tumor dataset extends this sequence of preclinical strength across prostate, lung, and breast models.
Market Pulse Summary
The stock dropped -10.9% in the session following this news. A negative reaction despite broadly favorable ATNM-400 data would fit the mixed pattern seen around prior preclinical and conference updates, where reactions ranged from -2.7% to double‑digit gains. Investors have at times discounted early-stage results or taken profits after earlier run‑ups. Past trading shows that even positive radiotherapy headlines have not guaranteed sustained strength, leaving room for volatility as the program advances toward clinical stages.
Key Terms
radioligand therapies medical
tyrosine kinase inhibitor medical
non-small cell lung cancer medical
antibody-drug conjugates medical
Actinium-225 medical
alpha emitter medical
double-stranded DNA breaks medical
interstitial lung disease medical
AI-generated analysis. Not financial advice.
- ATNM-400 demonstrates pan-tumor activity across prostate, lung, and breast cancer models, supporting multi-indication development potential
- In prostate cancer, ATNM-400 demonstrates efficacy across both high PSMA-expressing and, importantly, low PSMA-expressing prostate cancer models, unlike many PSMA-targeted radioligand therapies that work only in PSMA-high settings
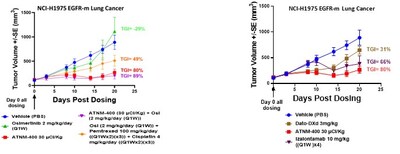
- In EGFR-mutant non-small cell lung cancer, ATNM-400 demonstrates greater tumor growth inhibition than osimertinib (a tyrosine kinase inhibitor) plus chemotherapy and outperforms the approved Trop-2 ADC Dato-DXd (DATROWAY®), the EGFR-cMET bispecific antibody amivantamab (RYBREVANT®) and the experimental EGFR-HER3 ADC izalontamab brengitecan, supporting potential use across first-, second-, and third-line treatment settings
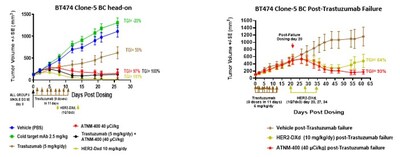
- In breast cancer, new head-to-head data shows ATNM-400 achieves efficacy comparable to the approved HER2-ADC trastuzumab deruxtecan (ENHERTU®) in trastuzumab-resistant models, with durable tumor control observed after treatment discontinuation - extending the Company's prior SABCS 2025 data and supporting potential for less frequent dosing of ATNM-400 compared to ADCs
- ATNM-400 is well tolerated, with no in vivo toxicities observed at efficacious doses, providing a favorable therapeutic index that supports monotherapy and combination development
ATNM-400 is a novel, first-in-class targeted radiotherapy whose differentiation stems from both its target and its isotope. The target is a non-PSMA membrane antigen associated with treatment resistance in advanced solid tumors that is overexpressed across prostate cancer, non-small cell lung cancer (NSCLC), and breast cancer, and is further upregulated following treatment with standard-of-care therapies — providing a strong mechanistic rationale for ATNM-400 in the treatment-resistant disease settings that represent the greatest unmet need, and for combination regimens designed to exploit this treatment-induced target upregulation. The isotope, Actinium-225 (Ac-225), is a potent alpha emitter that, compared to beta emitters such as Lu-177, delivers high-energy radiation capable of inducing irreversible double-stranded DNA breaks, with a shorter path length that may limit off-target effects and enhance therapeutic precision. Together, this target-and-isotope combination positions ATNM-400 to overcome conventional resistance pathways and deliver durable tumor control while potentially avoiding toxicities such as interstitial lung disease that limit the use of antibody-drug conjugates — expanding the population of patients who could benefit from treatment.
Key Data and Highlights From the ATNM-400 AACR Presentation
New preclinical data support ATNM-400 as a differentiated Ac-225 radioconjugate with potential applicability across multiple high-value solid tumor indications. ATNM-400 demonstrates a favorable tolerability profile, with no significant toxicity observed at therapeutic doses; and additionally:
In Prostate Cancer
- Demonstrates in vivo efficacy across prostate cancer models with low, medium, and high PSMA expression, including PSMA-negative models.
- Shows superior anti-tumor efficacy versus vehicle control, unconjugated antibody, and 177Lu–PSMA-617 (active ingredient in PLUVICTO®) in both high -PSMA, (C4-2) and low (22Rv1) PSMA-expressing models, addressing both patients unlikely to respond to PSMA-targeted radioligand therapy (low-PSMA, 22Rv1) and those who relapse on it (C4-2).
- Activity in PSMA-negative (DU145) models supports a differentiated profile, suggesting ATNM-400 could address mCRPC patients who are ineligible for or have progressed on PSMA-targeted radioligand therapy due to low or absent PSMA expression— a population with no currently approved targeted radiotherapy option.
In Lung Cancer
- New data in the NCI-H1975 EGFR-mutant NSCLC model - a clinically relevant model of osimertinib-resistant disease - shows ATNM-400 as monotherapy or in combination with osimertinib exceeds the tumor growth inhibition of osimertinib plus chemotherapy, the current standard of care in post-osimertinib progression. These results extend the Company's prior data demonstrating
100% complete tumor regression with the ATNM-400 plus osimertinib combination. - ATNM-400 monotherapy demonstrates greater anti-tumor activity than Dato-DXd (TROP-2 ADC approved in EGFR-mutant lung cancer) and izalontamab brengitecan (HER3-EGFR bispecific ADC in development for EGFR-mutant lung cancer). ATNM-400 also demonstrates greater anti-tumor activity than the EGFR-cMET bispecific antibody amivantamab (RYBREVANT®) as shown in prior studies.
In Breast Cancer
- New head-to-head data in the BT474 Clone-5 trastuzumab-resistant HER2+ breast cancer model which is a clinically relevant model of the post-trastuzumab setting, where treatment options are limited, demonstrate that ATNM-400, both as monotherapy and in combination with trastuzumab deruxtecan, achieves anti-tumor activity comparable to the approved HER2-ADC trastuzumab deruxtecan (ENHERTU®). These results extend the Company's SABCS 2025 data and position ATNM-400 as a potential alternative for patients who cannot tolerate HER2 ADCs due to interstitial lung disease, a known class-related toxicity.
- In the same post-trastuzumab failure setting, ATNM-400 produces durable tumor growth inhibition after treatment discontinuation which exceeds both vehicle control and trastuzumab deruxtecan, supporting the potential for less frequent dosing and more durable disease control than ADCs.
Sandesh Seth, Actinium's Chairman and CEO, said, "The data we presented at AACR are an important new piece of a much larger picture for ATNM-400. As a single agent, ATNM-400 continues to demonstrate activity across prostate, lung, and breast cancer in the treatment-resistant settings that represent the greatest unmet need, and also in combinations which can expand the available opportunity to additional patient populations. These data build on our previously disclosed results showing significant tumor regression when ATNM-400 is combined with osimertinib in EGFR-mutant lung cancer, and when combined with enzalutamide in prostate cancer, with similar combination potential emerging in breast cancer. What is becoming increasingly clear is that ATNM-400's target antigen is upregulated by standard-of-care therapies, which creates a strong mechanistic rationale for ATNM-400 to rescue patients who progress approved agents and also to extend the benefit of these approved agents through combinations. We look forward to continuing to advance ATNM-400 toward the clinic with additional data to come in 2026."
The ATNM-400 AACR presentation is available for viewing on the Presentations & Webinars page of Actinium's website HERE.
Title: Preclinical Development of ATNM-400, a First-in-Class Actinium-225 Radioconjugate with Pan-Tumor Efficacy in Solid Tumors
Abstract Number: 5824
About Actinium Pharmaceuticals, Inc.
Actinium is a pioneer in targeted radiotherapies designed to improve outcomes for patients with cancer. The company employs a biology-driven approach to develop differentiated radiopharmaceuticals for solid tumors and hematologic malignancies. Its mission is to transform cancer treatment through innovative radioconjugates that maximize therapeutic efficacy while minimizing toxicity to healthy tissue by combining expertise in tumor biology, translational medicine, and radiochemistry. Since inception, Actinium has focused on developing innovative radiotherapies. Its pipeline reflects this strategy across three areas: (1) solid tumor therapeutics including ATNM-400 and Actimab-A with pan-tumor potential; (2) Actimab-A as a therapeutic backbone for acute myeloid leukemia (AML) and myelodysplastic syndrome (MDS) in collaboration with the National Cancer Institute (NCI); and (3) targeted conditioning agents including Iomab-B for bone marrow transplant and Iomab-ACT for cell and gene therapy conditioning. ATNM-400 targets a novel antigen distinct from PSMA and has demonstrated preclinical activity across metastatic castration-resistant prostate cancer (mCRPC), non-small cell lung cancer (NSCLC), and breast cancer. Actimab-A has shown improved survival in relapsed/refractory AML with CLAG-M and is advancing toward a Phase 2/3 trial, with additional development ongoing through a CRADA with the NCI. Actinium is also advancing preclinical solid tumor programs and holds ~250 patents and patent applications, including intellectual property related to cyclotron-based production of Ac-225. For more information, please visit www.actiniumpharma.com.
For more information, please visit: https://www.actiniumpharma.com/
Forward-Looking Statements
This press release may contain projections or other "forward-looking statements" within the meaning of the "safe-harbor" provisions of the private securities litigation reform act of 1995 regarding future events or the future financial performance of the Company which the Company undertakes no obligation to update. These statements are based on management's current expectations and are subject to risks and uncertainties that may cause actual results to differ materially from the anticipated or estimated future results, including the risks and uncertainties associated with preliminary study results varying from final results, estimates of potential markets for drugs under development, clinical trials, actions by the FDA and other governmental agencies, regulatory clearances, responses to regulatory matters, the market demand for and acceptance of Actinium's products and services, performance of clinical research organizations and other risks detailed from time to time in Actinium's filings with the Securities and Exchange Commission (the "SEC"), including without limitation its most recent annual report on form 10-K, subsequent quarterly reports on Forms 10-Q and Forms 8-K, each as amended and supplemented from time to time.
Investors:
investorrelations@actiniumpharma.com
![]() View original content to download multimedia:https://www.prnewswire.com/news-releases/actinium-pharmaceuticals-inc-announces-compelling-pan-tumor-data-for-atnm-400-demonstrating-broad-efficacy-across-prostate-lung-and-breast-cancer-models-at-the-2026-american-association-of-cancer-research-annual-meeting-302749720.html
View original content to download multimedia:https://www.prnewswire.com/news-releases/actinium-pharmaceuticals-inc-announces-compelling-pan-tumor-data-for-atnm-400-demonstrating-broad-efficacy-across-prostate-lung-and-breast-cancer-models-at-the-2026-american-association-of-cancer-research-annual-meeting-302749720.html
SOURCE Actinium Pharmaceuticals, Inc.