Nevro Announces FDA Approval of HFX iQ™ Spinal Cord Stimulation System to Personalize the Treatment of Chronic Pain
Rhea-AI Summary
Nevro Corp. (NYSE: NVRO) announced FDA approval for its Senza HFX iQ spinal cord stimulation (SCS) system, the first AI-based SCS system designed to optimize pain relief through individualized patient data. This system aims to improve long-term pain management for conditions like chronic back pain and painful diabetic neuropathy. Key features include the HFX Algorithm, which processes over 20 million data points to personalize therapy. A limited U.S. launch is planned for late 2022, with broader availability expected in early 2023. European approval submissions are also underway.
Positive
- FDA approval for Senza HFX iQ enables market launch, potentially increasing revenue.
- The first AI-driven SCS system offers personalized pain management, enhancing patient care.
Negative
- Limited initial release may restrict market penetration and revenue generation.
- Approval is contingent on successful commercialization and addressing reimbursement challenges.
News Market Reaction
On the day this news was published, NVRO gained 5.02%, reflecting a notable positive market reaction.
Data tracked by StockTitan Argus on the day of publication.
Next Generation Senza® HFX iQ™ is the First and Only SCS System that Uses Artificial Intelligence to Optimize and Maintain Pain Relief Using Each Patient's Response
REDWOOD CITY, Calif., Oct. 12, 2022 /PRNewswire/ -- Nevro Corp. (NYSE: NVRO), a global medical device company that is delivering comprehensive, life-changing solutions for the treatment of chronic pain, today announced that it has received approval from the U.S. Food and Drug Administration (FDA) for the Senza HFX iQ spinal cord stimulation (SCS) system.
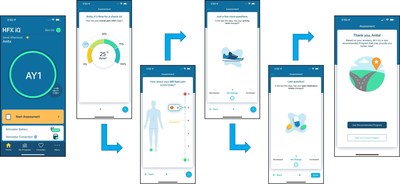
Senza HFX iQ is the first and only Artificial Intelligence-based SCS system that learns from patients. It was developed to address the variability in pain from patient to patient and to help patients optimize and maintain long-term pain relief and improved quality of life. The Senza HFX iQ system is comprised of the HFX iQ Implantable Pulse Generator (IPG), HFX Trial Stimulator, Charger and HFX App and will launch with algorithms specifically for treating chronic back and leg pain, including non-surgical back pain and painful diabetic neuropathy.
Senza HFX iQ is digitally-enabled and designed to optimize care by collecting patient data and guiding the patient through a customized treatment pathway. It provides several key advantages over other SCS systems:
- Patients start on the program most likely to provide pain relief, based on the HFX Algorithm™, which was built from more than 20 million datapoints and 80,000 implanted patients;
- Recommends customized therapy adjustments informed by patient inputs on the HFX App, including % pain relief, pain score, change in activity levels and change in pain medications;
- Directly adjusts the pain relief program on the patient's IPG from the HFX App on their smartphone, without the need for a separate manual remote;
- Captures patient and device data to enable sharing of patient-level outcomes with treating and referring physicians; and
- Provides a connected, digital environment around a patient's therapy that will allow for future updates to the HFX Algorithm, as well as new features and capabilities for patients and providers.
"HFX iQ is designed to improve the consistency of pain relief and is the only SCS system that truly personalizes care," stated D. Keith Grossman, Chairman and CEO of Nevro. "Pain is variable from patient to patient and over time. Using the big data from our HFX Cloud patient database, our unique HFX Algorithm was developed to identify those programs where patients have been more likely to get relief in the real world. HFX iQ takes direct input from each patient on their pain and quality of life measures to get smarter over time and recommend program changes. HFX iQ utilizes our superior high-frequency 10 kHz Therapy platform, which has been proven in clinical trials and is further supported by real-world evidence from more than 90,000 implanted patients. With HFX iQ, our goal is to provide physicians and patients the personalization needed to achieve and maintain the best possible long-term outcomes. We believe HFX iQ represents the future of SCS therapy."
Nevro will initiate a limited release of Senza HFX iQ in the U.S. this quarter, with a broad U.S. market launch planned for early 2023. In addition to the U.S. approval for HFX iQ, Nevro has submitted for approval in Europe.
Internet Posting of Information
Nevro routinely posts information that may be important to investors in the "Investor Relations" section of its website at www.nevro.com. The company encourages investors and potential investors to consult the Nevro website regularly for important information about Nevro.
About Nevro
Headquartered in Redwood City, California, Nevro is a global medical device company focused on delivering comprehensive, life-changing solutions that continue to set the standard for enduring patient outcomes in chronic pain treatment. The company started with a simple mission to help more patients suffering from debilitating pain and developed its proprietary 10 kHz Therapy™, an evidence-based, non-pharmacologic innovation that has impacted the lives of more than 90,000 patients globally. Nevro's comprehensive HFX™ spinal cord stimulation (SCS) platform includes a Senza SCS system and support services for the treatment of chronic pain of the trunk and limb and painful diabetic neuropathy.
Senza®, Senza II®, Senza Omnia™, and HFX iQ™ are the only SCS systems that deliver Nevro's proprietary 10 kHz Therapy™. Nevro's unique support services provide every patient with an HFX Coach™ throughout their pain relief journey and every physician with HFX Cloud™ insights for enhanced patient and practice management.
SENZA, SENZA II, SENZA OMNIA, OMNIA, HF10, the HF10 logo, 10 kHz Therapy, HFX, the HFX logo, HFX iQ, the HFX iQ logo, HFX Algorithm, HFX CONNECT, the HFX Connect logo, HFX ACCESS, the HFX Access logo, HFX COACH, the HFX Coach logo, HFX CLOUD, the HFX Cloud logo, RELIEF MULTIPLIED, the X logo, NEVRO, and the NEVRO logo are trademarks or registered trademarks of Nevro Corp. Patents covering Senza HFX iQ and other Nevro products are listed at Nevro.com/patents.
To learn more about Nevro, connect with us on LinkedIn, Twitter, Facebook and Instagram.
Forward-Looking Statements
In addition to historical information, this press release contains forward-looking statements reflecting the company's current beliefs and expectations of management made pursuant to the safe harbor provisions of the Private Securities Litigation Reform Act of 1995, including: our belief that HFX iQ represents the future of SCS therapy; and our expectation that we will initiate a limited launch of Senza HFX iQ in the U.S. this quarter, with a broad U.S. market launch planned for early 2023. These forward-looking statements are based upon information that is currently available to us or our current expectations, speak only as of the date hereof, and are subject to numerous risks and uncertainties, including our ability to successfully commercialize our products; our ability to manufacture our products to meet demand; the level and availability of third-party payor reimbursement for our products; our ability to effectively manage our anticipated growth and the costs and expenses of operating our business; our ability to protect our intellectual property rights and proprietary technologies; our ability to operate our business without infringing the intellectual property rights and proprietary technology of third parties; competition in our industry; additional capital and credit availability; our ability to attract and retain qualified personnel; and product liability claims. These factors, together with those that are described in greater detail in our Annual Report on Form 10-K filed on February 23, 2022, as well as any reports that we may file with the Securities and Exchange Commission in the future, may cause our actual results, performance or achievements to differ materially and adversely from those anticipated or implied by our forward-looking statements. We expressly disclaim any obligation, except as required by law, or undertaking to update or revise any such forward-looking statements.
Investors and Media:
Julie Dewey, IRC
Nevro Corp.
Chief Corp Communications and Investor Relations Officer
650-433-3247 | julie.dewey@nevro.com
![]() View original content to download multimedia:https://www.prnewswire.com/news-releases/nevro-announces-fda-approval-of-hfx-iq-spinal-cord-stimulation-system-to-personalize-the-treatment-of-chronic-pain-301647928.html
View original content to download multimedia:https://www.prnewswire.com/news-releases/nevro-announces-fda-approval-of-hfx-iq-spinal-cord-stimulation-system-to-personalize-the-treatment-of-chronic-pain-301647928.html
SOURCE Nevro Corp.