Quantum BioPharma Announces Peer-Reviewed Journal Publication of Groundbreaking Clinical Trial With Unbuzzd
Rhea-AI Summary
Quantum BioPharma (NASDAQ: QNTM) announced a peer‑reviewed publication (World Journal of Pharmaceutical and Medical Research, March 23, 2026) reporting a double‑blind, randomized, placebo‑controlled crossover trial of its dietary supplement unbuzzd.
Key findings: >40% faster reduction in blood alcohol concentration within 30 minutes, improved alertness and physiologic stability, 67% reduction in headache severity at four and eight hours, and no reported adverse side effects.
Positive
- BAC reduction >40% faster within 30 minutes
- Headache severity down 67% at 4 and 8 hours
- Statistically significant improvements vs placebo in alertness and physiologic measures
- Peer-reviewed publication in an international journal confirming clinical trial results
- No reported adverse effects in study participants
Negative
- Not an FDA drug approval; unbuzzd is described as a dietary supplement
- Does not permit driving; consuming unbuzzd after alcohol does not make operating a vehicle safe
- Individual results may vary, per company disclosure
News Market Reaction – QNTM
On the day this news was published, QNTM gained 38.39%, reflecting a significant positive market reaction. Argus tracked a peak move of +123.7% during that session. Our momentum scanner triggered 62 alerts that day, indicating high trading interest and price volatility. This price movement added approximately $5M to the company's valuation, bringing the market cap to $18M at that time. Trading volume was exceptionally heavy at 7.3x the daily average, suggesting very strong buying interest.
Data tracked by StockTitan Argus on the day of publication.
Key Figures
Market Reality Check
Peers on Argus
QNTM fell 5.08% while momentum peers like MNOV and INKT also moved down (median peer move about -4.2%), indicating broader Healthcare/Biotech pressure.
Previous Clinical trial Reports
| Date | Event | Sentiment | Move | Catalyst |
|---|---|---|---|---|
| Oct 02 | Lucid-MS study reports | Positive | +9.5% | Final 90-day toxicity and toxicokinetic reports support IND and planned Phase 2. |
| Oct 01 | Advisor appointment | Positive | +0.8% | MS expert Dr. Jack Antel joins as advisor for Lucid-MS clinical development. |
| Aug 11 | Manufacturing agreement | Positive | -10.1% | CDMO deal to manufacture oral Lucid-MS formulation for upcoming Phase 2 trial. |
| Aug 05 | Phase 1 MAD results | Positive | -11.6% | Phase 1 MAD trial shows Lucid-MS safety and tolerability in healthy participants. |
| May 28 | FSD202 Phase 2 greenlight | Positive | +1.5% | Ethics approval for Phase 2 FSD202 trial in MCAS-related nociplastic pain. |
Clinical and development milestones have produced mixed reactions, with both strong gains and notable selloffs around trial updates.
Over the past year, Quantum BioPharma has repeatedly highlighted its clinical pipeline. For Lucid-MS, it reported Phase 1 MAD safety, manufacturing preparations for an oral formulation, and final reports from 90‑day toxicity and toxicokinetic studies, all supporting a future IND and planned Phase 2 trials. It also secured ethics approval for a Phase 2 trial of FSD202 in MCAS-related nociplastic pain. Today’s peer‑reviewed publication for unbuzzd adds a separate, clinically supported product to this history of trial-driven announcements.
Historical Comparison
Past clinical-trial headlines for QNTM led to an average -1.99% move, showing that even positive study updates often met with cautious trading.
Clinical news has tracked steady progression: Lucid-MS advancing from Phase 1 safety to Phase 2-enabling work, while FSD202 moved into Phase 2 ethics-approved study design.
Market Pulse Summary
The stock surged +38.4% in the session following this news. A strong positive reaction aligns with Quantum BioPharma’s pattern of market interest in clinical milestones, as seen around prior Lucid-MS updates. However, the stock’s position 94.14% below its 52‑week high and past negative moves after some trial news suggest that enthusiasm could fade. Investors monitoring sustainability may focus on how this peer‑reviewed unbuzzd data integrates with the broader pipeline and past financing dynamics.
Key Terms
double-blind medical
randomized medical
placebo-controlled medical
crossover design medical
blood alcohol concentration medical
statistically significant technical
AI-generated analysis. Not financial advice.
unbuzzd Dramatically Accelerates Alcohol Metabolism, Speeds the Reduction of Blood Alcohol Concentration, and Reduces Symptoms of Intoxication, Impairment and Hangover
TORONTO, March 23, 2026 (GLOBE NEWSWIRE) -- Quantum BioPharma Ltd. (NASDAQ: QNTM) (CSE: QNTM) (FRA: 0K91) (“Quantum BioPharma” or the “Company”), "Company"), a biopharmaceutical company dedicated to building a portfolio of innovative assets and biotech solutions, announced the peer-reviewed publication of the double-blind, randomized, placebo-controlled crossover design clinical trial (NCT06505239) of its dietary supplement product, ‘unbuzzd’, comprising of a proprietary blend of vitamins and supplements developed by a world-class team of scientists at Quantum BioPharma. The trial, which investigated the effects of unbuzzd on alcohol intoxication and alcohol metabolism, was published here in the World Journal of Pharmaceutical and Medical Research (2026, volume 12, issue 3, pages 446-467).
The publication can be viewed by clicking the following link: https://zenodo.org/records/18873638
The clinical trial, designed to mirror real-life situations where people would consume unbuzzd after reaching an alcohol inebriated state included healthy male and female study participants between the ages of 21 and 43, with weights ranging from 119 to 232 pounds. Participants first drank measured quantities of alcohol to achieve a Blood Alcohol Concentration (“BAC”) between
Key findings from the clinical trial include:
- Accelerated Alcohol Metabolism: unbuzzd dramatically and rapidly reduced blood alcohol concentration in study participants. The rate at which BAC was lowered was on average for most participants more than 40 percent faster within the first 30 minutes of consuming unbuzzd compared to when participants consumed placebo.
- The faster reduction of BAC after consuming unbuzzd was statistically significant compared to the placebo and was observed in both males and females as shown in the figure below from the publication (note: FSD-F2R6 is the scientific name of the unbuzzd formulation). The continued acceleration of alcohol metabolism was also seen at subsequent BAC measurements over a four-hour period.
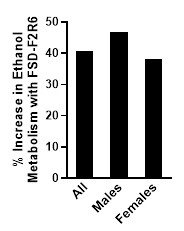
- Rapid Improvements in Alertness: Study participants reported statistically significant improvement in alertness as soon as 30 minutes after consuming unbuzzd. Participants felt more alert making fewer cognitive errors within 30 minutes of consuming unbuzzd, significantly outperforming placebo results.
- Rapid Improvements in Physiologic Changes due to Intoxication: unbuzzd lessened the elevation in heart rate and the drop in blood pressure that often accompanies alcohol intoxication, stabilizing both. This result was noted as statistically significant.
- Reduced Perceived Impairment and Mental Fatigue: unbuzzd helped alleviate perceived impairment and mental fatigue caused by alcohol intoxication.
- Hangover Relief: Participants in this study noted a statistically significant reduction in hangover symptoms. This included reduced cognitive and physical impairment, and reduced headache compared to placebo results, at both four hours (67 percent reduction in headache severity) and eight hours after consuming unbuzzd.
- No Side Effects: unbuzzd was well-tolerated by all study participants, with no reported adverse side effects.
“The unbuzzd clinical trial has now been published in a peer-reviewed international journal, and the trial confirms the exceptional overall efficacy of unbuzzd to help reduce the acute effects of alcohol intoxication and hangover, all without causing adverse side effects,” said Dr. Andrzej Chruscinski, Vice President of Clinical and Scientific Affairs at Quantum Biopharma. “We are thrilled to report this outcome.”
Anthony Durkacz, Founder and Executive Co-Chairman of Quantum BioPharma added, “The science and medical communities for centuries have believed there is no way to significantly speed up alcohol metabolism once it is in your bloodstream; your liver processes it at a largely constant rate of approximately one standard drink per hour, and that the only sobering agent is time. This publication validates unbuzzd as a responsible aid to reduce BAC, alleviate signs of alcohol intoxication, and assists with the dreaded hangover. Consumers can use unbuzzd knowing there is a peer-reviewed scientific publication with clinical trial evidence of the product’s efficacy.”
The formula for unbuzzd is licensed to Unbuzzd Wellness Inc. and is currently available at www.unbuzzd.com and www.amazon.com. unbuzzd is a registered trademark of Unbuzzd Wellness Inc.
Individual results may vary. unbuzzd is a dietary supplement. Consuming unbuzzd after alcohol ingestion does not permit you to operate a vehicle. Drink responsibly. DO NOT DRINK AND DRIVE.
About Unbuzzd Wellness Inc.
Unbuzzd Wellness inc., a non-trading but fully reporting public issuer, stands as a pioneering force in the wellness and recovery supplement landscape. unbuzzd has been developed by a world-class R&D team in pharmacology and medicine, with a commitment to innovation and quality. A proprietary blend of vitamins, minerals, and herbs, unbuzzd helps your body process alcohol faster, restore mental alertness, and improve cognition so you can drink responsibly and drink refreshingly. unbuzzd appeals to a broad target audience of alcohol consumers who want to have a good time, be in control, and still feel great the next day.
unbuzzd ready-to-mix powder sticks are available in 3-pack, 8-pack, and 18-pack formats at https://unbuzzd.com.
Individual results may vary. unbuzzd is a dietary supplement. Consuming unbuzzd after alcohol ingestion does not permit you to operate a vehicle. Drink responsibly. DO NOT DRINK AND DRIVE.

8-pack, 18-pack, and 3-pack formats of the unbuzzd ready-to-mix powder sticks
To view an enhanced version of this graphic, please visit: link
About Quantum BioPharma Ltd.
Quantum BioPharma is a biopharmaceutical company dedicated to building a portfolio of innovative assets and biotech solutions for the treatment of challenging neurodegenerative and metabolic disorders and alcohol misuse disorders with drug candidates in different stages of development. Through its wholly owned subsidiary, Lucid Psycheceuticals Inc. (“Lucid”), Quantum BioPharma is focused on the research and development of its lead compound, Lucid-MS. Lucid-MS is a patented new chemical entity shown to prevent and reverse myelin degradation, the underlying mechanism of multiple sclerosis, in preclinical models. Quantum BioPharma invented UNBUZZD™ and spun out its OTC version to a company, Unbuzzd Wellness Inc. (“UWI”), led by industry veterans. Quantum BioPharma retains ownership of
Forward-Looking Information
Certain information in this news release constitutes forward-looking statements under applicable securities laws. Any statements that are contained in this news release that are not statements of historical fact may be deemed to be forward-looking statements. Forward-looking statements are often identified by terms such as “may”, “should”, “anticipate”, “expect”, “potential”, “believe”, “intend” or the negative of these terms and similar expressions.
Readers are cautioned that the foregoing list is not exhaustive. Readers are further cautioned not to place undue reliance on forward-looking statements, as there can be no assurance that the plans, intentions or expectations upon which they are placed will occur. Such information, although considered reasonable by management at the time of preparation, may prove to be incorrect and actual results may differ materially from those anticipated.
Forward-looking statements contained in this press release are expressly qualified by this cautionary statement and reflect the Company’s expectations as of the date hereof and are subject to change thereafter. The Company undertakes no obligation to update or revise any forward-looking statements, whether as a result of new information, estimates or opinions, future events or results or otherwise or to explain any material difference between subsequent actual events and such forward- looking information, except as required by applicable law.
Contacts:
Quantum BioPharma Ltd.
Zeeshan Saeed, Founder, CEO and Executive Co-Chairman of the Board
Email: Zsaeed@quantumbiopharma.com
Telephone: (416) 854-8884
Investor Relations
Email: ir@quantumbiopharma.com, info@quantumbiopharma.com
Website: www.quantumbiopharma.com
Photos accompanying this announcement are available at:
https://www.globenewswire.com/NewsRoom/AttachmentNg/600ca40f-b94f-49bc-8d5f-5ff43576b965
https://www.globenewswire.com/NewsRoom/AttachmentNg/213c2b0f-182e-4ac0-b562-031c143ed6cf

FAQ
What did Quantum BioPharma (QNTM) announce on March 23, 2026 about unbuzzd?
How much did unbuzzd speed blood alcohol concentration (BAC) reduction in the QNTM study?
Did the QNTM clinical trial report improvements in hangover and symptoms of intoxication?
Were there safety concerns reported in the March 23, 2026 QNTM unbuzzd trial?
Can consumers drive after taking unbuzzd following alcohol consumption, per Quantum BioPharma (QNTM)?







